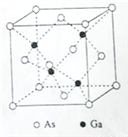
��Ŀ����
��������ʵ��������������õ��Ľ�����ȷ���ǣ� )
ѡ�� | ʵ����������� | ���� |
A | ���������Һ�еμ�1�η�̪��Ȼ����μ���ϡ��������ɫ��ȥ��2min���Թ���������� | �ǽ����ԣ�Cl>Si |
B | ����м��ȵ�AgNO3��Һ�еμ�KCl��Һ����Һ�ɺ�ɫ��Ϊ��ɫ | KCl��Һ���м��� |
C | ��CuSO4��Һ�еμ�KI��Һ���ټ��뱽�����а�ɫ�������ɣ��������ɫ | ��ɫ��������ΪCuI |
D | ij��Һ�μ������ữ��BaCl2��Һ�����ɰ�ɫ���� | ����Һ�п��ܲ���SO42- |
A. A B. B C. C D. D
��ϰ��ϵ�д�
�����Ŀ



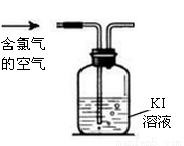

 ��
�� ��
��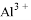 ���������ӣ��������м��������
���������ӣ��������м��������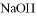 ��Һ���Ȳ����裬���ˣ����������ټ�����������ᣬ������Һ�д������ӵ��������ǣ� ��
��Һ���Ȳ����裬���ˣ����������ټ�����������ᣬ������Һ�д������ӵ��������ǣ� ��