��Ŀ����
(1)����ʵ��������ʵ����ʵ��������ȷ����________(�����)�������Թ��еμ�Һ��ʱ��Ϊ��ʹҺ��ε��Թ���Ӧ����ͷ�ι������Թ��У�
������100 mL 1.00mol/L��NaCl��Һʱ������������ƽ��ȡ5.85 gNaCl���壻
����100��ʱ��NaOHϡ��Һ�еμӱ��͵�FeCl3��Һ�����Ʊ�Fe(OH)3���壻
�������Ȼ�����Һʱ���Ƚ��Ȼ�����������������ܽ⣬Ȼ���ˮϡ�ͣ�
�ݷ�Һ©����Һʱ���Ƚ��²��Һ����¿�������Ȼ���ٴ��¿������ϲ��Һ��
�ⶨij��ҺpHʱ��ȡһС��������ֽ���벣��Ƭ�ϣ��ò�����պȡ��Һ��
����ֽ���в�������pH��ֽ��ɫ���Ա�(2)����ͼ��ʾ��ʵ��װ���У�ʢ������ˮ��ˮ����������ձ���С�ձ����������ͭƬ����Ũ���ᣬС�ձ����浹��һ���ձ�����ش��������⣺

��.ʵ������У��۲쵽����Ҫ�����ǣ�
��ͭƬ����������ݣ�ͭƬ��С������ʧ��
��С�ձ�����Һ����ɫ�����ɫ��
��________________________________________________��
��________________________________________________��
��.�ø�װ����ͭ��Ũ���ᷴӦ��ʵ�飬��ͻ�����ŵ���______________��
��.��Ҫ��֤���յõ����������������ķ�����______________��
(1)�ܢ�
(2)I �۴��ձ����к���ɫ�������ɣ����������ɫ������ȥ �ܴ��ձ���Һ��������ˮ����Һ���½�
��.��Ӧ��ϵ��գ����������ݳ�������Ⱦ����
��.�������۵Ĵ��ձ������������Ŀ��������к���ɫ�������ɣ���֤����������������ΪNO����

 ����νӽ̲���ĩ���Ԥϰ�人������ϵ�д�
����νӽ̲���ĩ���Ԥϰ�人������ϵ�д� ������ҵ��ٳɳ����½������������ϵ�д�
������ҵ��ٳɳ����½������������ϵ�д�(12��)(1)����ʵ��������ʵ����ʵ��������ȷ����__(�����)
| A����������ƽ��ȡ4.00g�����NaOH���壬�ܽ���6ml����ˮ�У�����10��NaOH��Һ������������ͭ����Һ����һ��������ͭ��Һ�У���������10������������Һ�� |
| B��ʵ��������ʯ��ʱ����������ƿ�м���һЩ���Ƭ����ֹ���У��������¿ڽ���ˮ���Ͽڳ���ˮ�� |
| C�����ǻ��ý������Ϳ�����������ˮ��Ӧ��ʵ���ҽ��Ʊ�������ˮ�ƾ��С� |
| D��ú����������л�����ɵĸ��ӻ������к��б����ױ������ױ����л�������ø�����ñ��� |
F������������������л��е�NaN03���ʣ������������ᴿ�����ʡ�
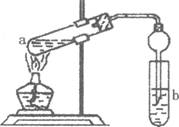
(2)����ͼ�����Թ�a���ȼ���2mL95�����Ҵ�����ҡ����������5mLŨH2S04�����ҡ�ȣ���ȴ���ټ���2g��ˮ�����ƣ��ò�������ֽ�����Թ̶ܹ�������̨�ϣ����Թ�b�м��� 7mL����̼������Һ�����Ӻ�װ�á��þƾ��ƶ��Թ�a���ȣ����۲쵽�Թ�b������������ʱֹͣʵ�顣
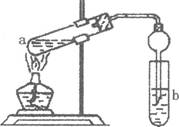
�ټ���ŨH2S04��������______________________________________
���Թ�b�й۲쵽����Ҫ������_________________________________
����ʵ�������θ���ܳ������������⣬��һ����Ҫ������___________________________
�ܱ���Na2C03��Һ��������__________________________________