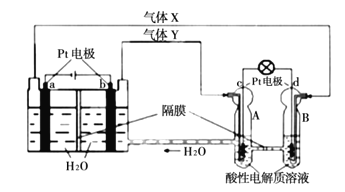
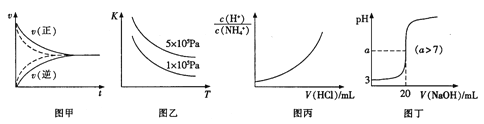
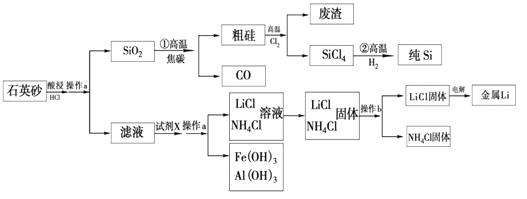
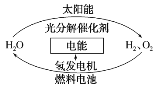
��Ŀ����
����Ŀ��һ���¶��£�������Һ������Ũ�ȹ�ϵʽ��ȷ����( )
A. pH��5��H2S��Һ�У�c(H��)��c(HS��)��1��10��5mol��L��1
B. pH��a�İ�ˮ��Һ��ϡ��10������pH��b����a��b��1
C. �����£�0.1mol/LpH=7.82��NH4HCO3��Һ�У�c��HCO3-����c��NH4+����c��OH-����c��H+��
D. pH��ͬ�Ģ�CH3COONa ��NaHCO3 ��NaClO������Һ��c(Na��)���٣��ڣ���
���𰸡�D
��������
A. pH��5��H2S��Һ�У�c(H��)��1��10��5mol��L��1�����ݵ���غ�c(H��)�Tc��OH-��+2c(S2-)+c(HS-)����֪��c(H��)> c(HS-)����c(HS-)<1��10��5mol��L��1����A������
B.һˮ�ϰ�Ϊ������������ڵ���ƽ������pH=a�İ�ˮ��Һϡ��10������һˮ�ϰ��ĵ���̶�������ϡ�ͺ���Һ��pH�ı仯С��1����a<b��1����B������
C. 0.1mol/L��NH4HCO3��Һ��pH��7.82��˵����Һ�Լ��ԣ���笠���ˮ��̶�С��̼�������ˮ��̶ȣ���c��NH4+����c��HCO3-������C������
D.pH��ͬ�Ģ�CH3COONa ��NaHCO3 ��NaClO������Һ��������CH3COOH>H2CO3>HClO ����ˮ��̶���![]() ��
��![]() �ۣ�pH��ͬʱ������Һ��Ũ����
�ۣ�pH��ͬʱ������Һ��Ũ����![]() ��
��![]() �ۣ���������Һ��c(Na��)���٣��ڣ��ۣ�����Dѡ������ȷ����
�ۣ���������Һ��c(Na��)���٣��ڣ��ۣ�����Dѡ������ȷ����
��ѡD��