��Ŀ����
����Ŀ���������ʯ��ʯī��Ϊ̼��ͬ�������壬��������������ʱȼ������һ����̼������������ʱ���ȼ�����ɶ�����̼����Ӧ�зų���������ͼ��ʾ��
��1����ͨ��״����,���ʯ��ʯī��Ƚ�, ���������ʯ������ʯī�������ȶ���ʯī��ȼ����Ϊ ��
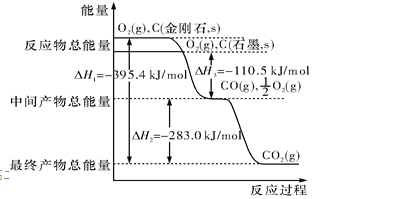
��2��12 gʯī��һ����������ȼ��,��������36 g���ù��̷ų�������Ϊ ��
��������������ȼ�գ������������ֹ�̬������P2O3��P2O5 �� P2O5��ͨ��״̬���ȶ�����֪3.1 g�ĵ�������P����3.2 g������ȼ�գ�����Ӧ��ľ����ų�X kJ������
��3��д���������������Ӧ��������g��Ϊ �� ��
��4����֪������ȼ����ΪY kJ/mol��д��1mol P��O2��Ӧ���ɹ�̬P2O3���Ȼ�ѧ����ʽ ��
���𰸡�
��1��ʯī��-393.5kJ/mol
��2��252.0kJ
��3��P2O3��2.75g; P2O5��3.55g��
��4��-��20X-Y��kJ/mol
��������
�����������1��ͼ��������ʯ��������ʯī������Խ��Խ�ȶ�������˵��ʯī�ȶ���ͼ�����1molʯī��ȫȼ������1mol������̼�ų�������Ϊ393.5kJ����ʯī��ȼ����Ϊ393.5kJmol-1���ʴ�Ϊ��ʯī��393.5kJmol-1��
��2��12gʯī���ʵ���Ϊ1mol����һ����������ȼ�գ�����Ԫ���غ㣬�����ɶ�����̼����Ϊ44g��������һ����̼����Ϊ28g����������36g��28g��36g��44g���ж����ɵ�����Ϊһ����̼�Ͷ�����̼���壬��һ����̼���ʵ���Ϊx��������̼���ʵ���Ϊ��1-x��mol��28x+44��1-x��=36g��x=0.5mol��������̼���ʵ���Ϊ0.5mol������ͼ�������C��ʯī��s��+O2��g���TCO2��g����H=-393.5 kJmol-1 ��C��ʯī��s��+![]() O2��g���TCO��g����H=-110.5 kJmol-1 �����ɶ�����̼��һ����̼�������ų�����=393.5 kJmol-1 ��0.5mol+110.5 kJmol-1 ��0.5mol=252KJ��12gʯī��һ����������ȼ�գ���������36g���ù��̷ų�������Ϊ252.0kJ���ʴ�Ϊ��252.0 kJ��
O2��g���TCO��g����H=-110.5 kJmol-1 �����ɶ�����̼��һ����̼�������ų�����=393.5 kJmol-1 ��0.5mol+110.5 kJmol-1 ��0.5mol=252KJ��12gʯī��һ����������ȼ�գ���������36g���ù��̷ų�������Ϊ252.0kJ���ʴ�Ϊ��252.0 kJ��
��3��3.1g�ĵ�������P�������ʵ���Ϊ![]() =0.1mol��3.2g�����������ʵ���Ϊ
=0.1mol��3.2g�����������ʵ���Ϊ![]() =0.1mol����Pԭ����Oԭ�ӵ���Ŀ֮��Ϊ0.1mol��0.1mol��2=1��2��2��5��1��2��2��3���ʷ�Ӧ����ΪP2O3��P2O5�������ʵ����ֱ�Ϊxmol��ymol����2x+2y=0.1 ����3x+5y=0.1��2 �������٢��ã�x=0.025mol��y=0.025mol����P2O3������Ϊ0.025mol��110g/mol=2.75g��P2O5������Ϊ0.025mol��142g/mol=3.55g���ʴ�Ϊ��P2O3��P2O5��2.75g��3.55g��
=0.1mol����Pԭ����Oԭ�ӵ���Ŀ֮��Ϊ0.1mol��0.1mol��2=1��2��2��5��1��2��2��3���ʷ�Ӧ����ΪP2O3��P2O5�������ʵ����ֱ�Ϊxmol��ymol����2x+2y=0.1 ����3x+5y=0.1��2 �������٢��ã�x=0.025mol��y=0.025mol����P2O3������Ϊ0.025mol��110g/mol=2.75g��P2O5������Ϊ0.025mol��142g/mol=3.55g���ʴ�Ϊ��P2O3��P2O5��2.75g��3.55g��
��4��������ȼ����ΪYkJ/mol����1mol����ȫȼ�����ɹ�̬P2O5�ų�������ΪYkJ����Ӧ������0.025molP2O5����Ҫ��0.05mol����0.05mol��ȼ���������ɹ�̬P2O5�ų�������Ϊ0.05YkJ����Ӧ������0.025molP2O3����Ҫ��0.05mol������0.05mol��ȼ���������ɹ�̬P2O3�ų�������ΪXkJ-0.05YkJ=��X-0.05Y��kJ������1molP��O2��Ӧ���ɹ�̬P2O3�ų�������Ϊ��X-0.05Y��kJ��![]() =��20X-Y��kJ����1mol P��O2��Ӧ���ɹ�̬P2O3�ķ�Ӧ����H=-��20X-Y��kJ/mol���ʴ�Ϊ��-��20X-Y��kJ/mol��
=��20X-Y��kJ����1mol P��O2��Ӧ���ɹ�̬P2O3�ķ�Ӧ����H=-��20X-Y��kJ/mol���ʴ�Ϊ��-��20X-Y��kJ/mol��

 ��У����ϵ�д�
��У����ϵ�д�