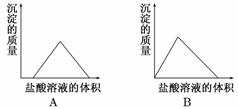
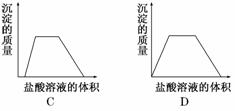
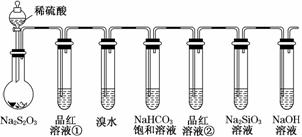
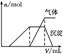
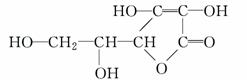
��Ŀ����
��FeCl3��Һʴ��ͭ�������·��Ĺ����У���Һ��������Դ���յĹ��̼������£�
�����Һ��Ͷ�������м����ַ�Ӧ�������������Һ��
������Һ�м���һ����ʯ��ˮ��������ҺpH��ͬʱ���������Ŀ�����
��֪��Ksp[Fe(OH)3]��4.0��10��38
�ش��������⣺
(1)FeCl3ʴ��ͭ����Ӧ�����ӷ���ʽΪ________________________________________��
(2)���̢������м����Ҫ������__________________������õ��������Ҫ�ɷ���________���ӹ����з����ͭ����õķ�����___________________________________��
(3)���̢��з�����Ӧ�Ļ�ѧ����ʽΪ________________________________��
(4)���̢��е�����Һ��pHΪ5����������Ũ��Ϊ
________________________________________________________________________
________________________________________________________________________��(��ʽ����)
������(1)Fe3������ǿ�����ԣ��ܽ�Cu������Cu2����
(2)��Һ�к���Cu2�������̢��м��������м������Ӧ��Cu2����Fe===Fe2����Cu��������Ĺ�����ͭ�������������ܽ������ɷ����ͭ��
(3)���̢����Һ�к���FeCl2������ʯ��ˮ������Ӧ��FeCl2��Ca(OH)2===Fe(OH)2����CaCl2������ ������ɽ�Fe(OH)2����ΪFe(OH)3��
������ɽ�Fe(OH)2����ΪFe(OH)3��
(4)��Ksp[Fe(OH)3]��pH��5��֪��c(Fe3��)��c3(OH��)��c(Fe3��)��(10��9)3��4.0��10��38����c(Fe3��)��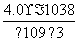 ��4.0��10��11 mol/L��
��4.0��10��11 mol/L��
�𰸣�(1)2Fe3����Cu===2Fe2����Cu2��
(2)����ͭ��ͭ����������������
(3)FeCl2��Ca(OH)2===Fe(OH)2����CaCl2
4Fe(OH)2��O2��2H2O===4Fe(OH)3
(4)c(Fe3��)��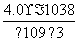 ��4.0��10��11 mol/L
��4.0��10��11 mol/L

�������������������γ��������Ҫ���ʡ�ij�������п��ܺ����������ӣ�Na����Ba2����NH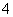 ��Al3����Cl����SO
��Al3����Cl����SO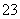 ��SO
��SO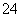 ��NO
��NO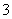 �ȡ�ij�о�С��ȡ�õ�һ���������꣬Ũ�������ó�����Һ�ֳ����ݣ���������ʵ�飺
�ȡ�ij�о�С��ȡ�õ�һ���������꣬Ũ�������ó�����Һ�ֳ����ݣ���������ʵ�飺
| ���� | �����Լ� | ʵ������ |
| ��һ����Һ | �μ������ĵ���KI��Һ | ��Һ����ɫ |
| �ڶ�����Һ | �μ��������ữ��BaCl2��Һ | �а�ɫ�������� |
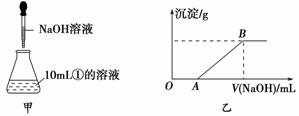
| ��������Һ | �μ�NaOH��Һ�����ȣ������NaOH��Һ���(V)�����ɵij�������������������ʵ���(n)�Ĺ�ϵ����ͼ |
|
��ش��������⣺
(1)����ʵ�����жϸ������п϶������ڵ�������______________������ȷ����������________________��
(2)д����һ����Һ�μӵ���KI��Һʱ������Ӧ�����ӷ���ʽ��__________________��
(3)��������Һ�μ�NaOH��Һ�����ȣ����������з����˶����Ӧ��д������������Ӧ�����ӷ���ʽ��__________________________________��__________________________��
(4)���ʵ�鷽����������������Ƿ����Cl����___________________________________
______________________________��
(5)��С��Ϊ��̽��NO����������������γɹ��̣�����ƿ�г��뺬������NO��SO2���壬������ͨ��O2��������ѧ��Ӧ����������������ˮ�������������꣬��NO���� ����Ӧ�е�������________________________________________________________��
����Ӧ�е�������________________________________________________________��
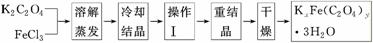
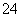 ת��ΪH2C2O4����0.100 0 mol��L��1KMnO4��Һ�ζ���������KMnO4��Һ24.00 mLʱǡ����ȫ��Ӧ��H2C2O4������KMnO4��Һ��Ӧ�Ļ�ѧ����ʽ�ǣ�__________________________��������Һ�м��������Ļ�ԭ����ǡ�ý�Fe3����ȫת��ΪFe2������KMnO4��Һ�����ζ�����Fe2����ȫ����ʱ����ȥKMnO4��Һ4.00 mL���˵ζ���Ӧ�����ӷ���ʽ��____________��
ת��ΪH2C2O4����0.100 0 mol��L��1KMnO4��Һ�ζ���������KMnO4��Һ24.00 mLʱǡ����ȫ��Ӧ��H2C2O4������KMnO4��Һ��Ӧ�Ļ�ѧ����ʽ�ǣ�__________________________��������Һ�м��������Ļ�ԭ����ǡ�ý�Fe3����ȫת��ΪFe2������KMnO4��Һ�����ζ�����Fe2����ȫ����ʱ����ȥKMnO4��Һ4.00 mL���˵ζ���Ӧ�����ӷ���ʽ��____________��