��Ŀ����
����ʵ�鷽���У����ܴﵽʵ��Ŀ�ĵ���
ѡ�� | ʵ��Ŀ�� | ʵ�鷽�� |
A | ����CH3CH2Br��NaOH��Һ���Ƿ���ˮ�� | ��CH3CH2Br��NaOH��Һ���ȡ���ȴ��ȡ���ϲ�ˮ��Һ����ϡHNO3�ữ������AgNO3��Һ���۲��Ƿ��������ɫ���� |
B | ����Fe(NO3)2�����Ƿ����������� | ��Fe(NO3)2��Ʒ����ϡH2SO4�μ�KSCN��Һ���۲���Һ�Ƿ��� |
C | ��֤Br2��������ǿ��I2 | ��������ˮ����KI��Һ�У��ټ���CCl4�������ã��ɹ۲쵽�²�Һ�����ɫ |
D | ��֤AgI���ܽ��С��AgCl | ��NaIŨ��Һ����AgCl����Һ�У����ɹ۲쵽�����ɰ�ɫ��Ϊ��ɫ |
A. A B. B C. C D. D
 ͬ����ϰǿ����չϵ�д�
ͬ����ϰǿ����չϵ�д�������(CH3OCH3)����Ϊ21���͵�����ȼ�ϣ���CO��H2Ϊԭ��������������Ҫ��������������Ӧ��
��ѧ��Ӧ����ʽ | ��ѧƽ�ⳣ�� | |
��CO(g)��2H2(g) | ��H1=-99 kJ•mol-1 | K1 |
��2CH3OH(g) | ��H2����24 kJ•mol-1 | K2 |
��CO(g)��H2O(g) | ��H3����41 kJ•mol-1 | K3 |
��1���ù��յ��ܷ�ӦΪ3CO(g)��3H2(g) CH3OCH3(g)��CO2(g) ��H
CH3OCH3(g)��CO2(g) ��H
�÷�Ӧ��H��__________________����ѧƽ�ⳣ��K��____________________(�ú�K1��K2��K3�Ĵ���ʽ��ʾ)��
��2��ij�¶��£���8.0molH2��4.0molCO�����ݻ�Ϊ2L���ܱ������У�������Ӧ��4H2(g)+2CO(g)  CH3OCH3(g)+H2O(g)��10 ���Ӻ�Ӧ��ƽ�⣬��ö����ѵ��������Ϊ25%����CO��ת����Ϊ________��
CH3OCH3(g)+H2O(g)��10 ���Ӻ�Ӧ��ƽ�⣬��ö����ѵ��������Ϊ25%����CO��ת����Ϊ________��
��3�����д�ʩ�У������CH3OCH3���ʵ���________��
A������������� B�������¶� C�����ø�Ч���� D������ѹǿ
��4���ù����з�Ӧ�۵ķ��������CH3OCH3�IJ��ʣ�ԭ����_______________________________��


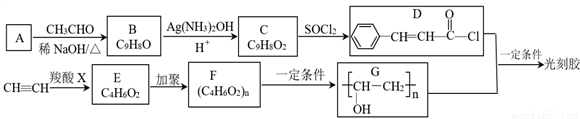

 CH��RCOOCH=CH2
CH��RCOOCH=CH2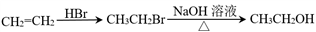 ��
�� 2AB ��H= Q kJ/mol�ķ�Ӧʱ����������AB������������AB%���Ĺ�ϵ��ͼ��ʾ������a��bΪ��ͬ�¶�ʱ�ķ�Ӧ���ߣ������������䣩��cΪ��t3ʱ�̿�ʼ��С�����ݻ����¶Ȳ��䣩�ı仯���ߡ������й�˵����ȷ����
2AB ��H= Q kJ/mol�ķ�Ӧʱ����������AB������������AB%���Ĺ�ϵ��ͼ��ʾ������a��bΪ��ͬ�¶�ʱ�ķ�Ӧ���ߣ������������䣩��cΪ��t3ʱ�̿�ʼ��С�����ݻ����¶Ȳ��䣩�ı仯���ߡ������й�˵����ȷ����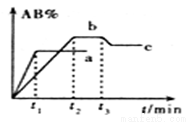
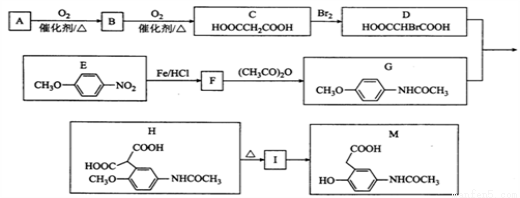

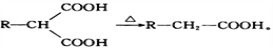
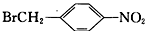 Ϊԭ�ϣ����Լ���ѡ��������Ʊ�
Ϊԭ�ϣ����Լ���ѡ��������Ʊ�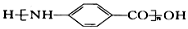 �ĺϳ�·�ߣ�_______________________________________________��
�ĺϳ�·�ߣ�_______________________________________________��