��Ŀ����
 ��2009?����ģ�⣩�״�������ˮ������Ӧ������������Ӧ����ʽ���£�
��2009?����ģ�⣩�״�������ˮ������Ӧ������������Ӧ����ʽ���£�CH3OH��g��+H2O��g��?CO2��g��+3H2��g������H��0
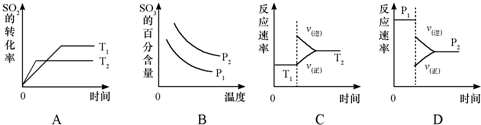
��1��һ�������£������Ϊ2L�ĺ������ܱ������г���1molCH3OH��g����3molH2O��g����20s��û�������ѹǿ�Ƿ�Ӧǰ��1.2�������ü״���ʾ�÷�Ӧ������Ϊ
0.01mol/L?s
0.01mol/L?s
����2���жϣ�1���п��淴Ӧ�ﵽƽ��״̬�������ǣ�����ţ�
�ۢ�
�ۢ�
����v����CH3OH��=v����CO2��
�ڻ��������ܶȲ���
�ۻ�������ƽ����Է�����������
��CH3OH��H2O��CO2��H2��Ũ�ȶ����ٷ����仯
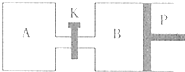
��3����ͼ��P�ǿ�����ƽ�л����Ļ������ر�K������ͬ�¶�ʱ����A�����г���1molCH2OH��g����2molH2O��g������B�����г���1.2molCH3OH��g����2.4molH2O��g�����������ֱ���������Ӧ��
��֪��ʼʱ����A��B�������ΪaL���Իش�
�ٷ�Ӧ�ﵽƽ��ʱ����B�����Ϊ1.5aL������B��CH3OHת����Ϊ
75%
75%
A��B��������H2O��g��������ٷֺ����Ĵ�С��ϵΪ��B�������������������=������
��
A��������K��һ��ʱ������´ﵽƽ�⣬����B�����Ϊ
1.75a
1.75a
L����ͨ��������������Բ��ƣ��Ҳ������¶ȵ�Ӱ�죩����������1�����ݻ�ѧƽ������ʽ��ʽ���㣬����ѹǿ֮�ȵ������ʵ���֮�ȣ�
��2����ѧƽ��ı�־�����淴Ӧ������ͬ�������Ũ�ȱ��ֲ������ѡ�
��3�������ݺ��º��ݡ����º�ѹ�����仯����Ϸ�Ӧƽ�����������жϣ�ѹǿ�仯�����ʵ����仯�����ȣ�
�ڴ�KAB��ɵ��Ǻ��º�ѹ����������ѹǿ�仯�����ʵ����仯�����ȼ���õ���
��2����ѧƽ��ı�־�����淴Ӧ������ͬ�������Ũ�ȱ��ֲ������ѡ�
��3�������ݺ��º��ݡ����º�ѹ�����仯����Ϸ�Ӧƽ�����������жϣ�ѹǿ�仯�����ʵ����仯�����ȣ�
�ڴ�KAB��ɵ��Ǻ��º�ѹ����������ѹǿ�仯�����ʵ����仯�����ȼ���õ���
����⣺��1�����ݻ�ѧƽ������ʽ��ʽ���㣬�跴Ӧ�ļ״����ʵ���Ϊx
CH3OH��g��+H2O��g��?CO2��g��+3H2��g��
��ʼ����mol�� 1 3 0 0
�仯����mol�� x x x 3x
20s�� ��mol�� 1-x 3-x x 3x
20s��û�������ѹǿ�Ƿ�Ӧǰ��1.2����4+2x=1.2��4
x=0.4mol
v��CH3OH��=
=0.01mol/L?s��
�ʴ�Ϊ��0.01mol/L?s��
��2�����淴Ӧ�ﵽƽ��״̬�����������淴Ӧ������ͬ��
��v����CH3OH��=v����CO2���Ƿ�Ӧ����֮�ȵ��ڻ�ѧ����ʽ������֮�ȣ�����˵�㷴Ӧ�ﵽƽ�⣬�ʢٲ����ϣ�
�ڷ�Ӧǰ�������������䣬����������䣬��Ӧ�������ܶȲ��䣬���������ܶȲ��䲻��˵����Ӧ�ﵽƽ�⣬�ʢڲ����ϣ�
�۷�Ӧǰ���������ʵ������������غ㣬��������ƽ����Է����������䣬˵����Ӧ�ﵽƽ��״̬���ʢ۷��ϣ�
��CH3OH��H2O��CO2��H2��Ũ�ȶ����ٷ����仯��ƽ���־���ʢܷ��ϣ�
�ʴ�Ϊ���ۢܣ�
��3����B�����������Ϊԭ����1.5�������ӵ���������ʵ���Ϊ3.6 mol��0.5=1.8 mol�����ݻ�ѧ����ʽ��֪����2mol����Ӧ�״�1mol����Ӧ�ļ״������ʵ���Ϊ0.9 mol��CH3OH��ת����=
��100%=75%��A�Ǻ��ݷ�Ӧ������B�Ǻ�ѹ��Ӧ������B��������1.2molCH3OH��g�� ��2.4molH2O��g����B��������1molCH2OH��g����2molH2O��g�����ǵ�Чƽ�⣮��Ӧ����ʽ���������������࣬��ӦʱA����ѹǿ��B����������A��ƽ��ʱ�൱��Bƽ���ļ�ѹ����ѹƽ�����淴Ӧ�����ƶ���A������H2O�ٷֺ������࣬��B����A����
�ʴ�Ϊ��75%������
�ڴ�Kʱ��AB���һ�����µ�ѹ��������Ӧ����ʼͶ�������ʵ�����ƽ�������������ȣ����K���´ﵽƽ����ܵ����Ϊx����x����3+3.6��=1.5a��3.6
���x=2.75a������B�����Ϊ2.75a-a=1.75a��
�ʴ�Ϊ��1.75a��
CH3OH��g��+H2O��g��?CO2��g��+3H2��g��
��ʼ����mol�� 1 3 0 0
�仯����mol�� x x x 3x
20s�� ��mol�� 1-x 3-x x 3x
20s��û�������ѹǿ�Ƿ�Ӧǰ��1.2����4+2x=1.2��4
x=0.4mol
v��CH3OH��=
| ||
| 20s |
�ʴ�Ϊ��0.01mol/L?s��
��2�����淴Ӧ�ﵽƽ��״̬�����������淴Ӧ������ͬ��
��v����CH3OH��=v����CO2���Ƿ�Ӧ����֮�ȵ��ڻ�ѧ����ʽ������֮�ȣ�����˵�㷴Ӧ�ﵽƽ�⣬�ʢٲ����ϣ�
�ڷ�Ӧǰ�������������䣬����������䣬��Ӧ�������ܶȲ��䣬���������ܶȲ��䲻��˵����Ӧ�ﵽƽ�⣬�ʢڲ����ϣ�
�۷�Ӧǰ���������ʵ������������غ㣬��������ƽ����Է����������䣬˵����Ӧ�ﵽƽ��״̬���ʢ۷��ϣ�
��CH3OH��H2O��CO2��H2��Ũ�ȶ����ٷ����仯��ƽ���־���ʢܷ��ϣ�
�ʴ�Ϊ���ۢܣ�
��3����B�����������Ϊԭ����1.5�������ӵ���������ʵ���Ϊ3.6 mol��0.5=1.8 mol�����ݻ�ѧ����ʽ��֪����2mol����Ӧ�״�1mol����Ӧ�ļ״������ʵ���Ϊ0.9 mol��CH3OH��ת����=
| 0.9mol |
| 1.2mol |
�ʴ�Ϊ��75%������
�ڴ�Kʱ��AB���һ�����µ�ѹ��������Ӧ����ʼͶ�������ʵ�����ƽ�������������ȣ����K���´ﵽƽ����ܵ����Ϊx����x����3+3.6��=1.5a��3.6
���x=2.75a������B�����Ϊ2.75a-a=1.75a��
�ʴ�Ϊ��1.75a��
���������⿼���˻�ѧƽ������ʽ���㣬ƽ���־�ķ����жϣ����º������������º�ѹ�����ķ����ж��ǽ���ؼ�������ѹǿ֮�Ⱥ����ʵ���֮�ȳ�����������Ŀ�ѶȽϴ�

��ϰ��ϵ�д�
 �������¿��ÿ�ʱ��ҵϵ�д�
�������¿��ÿ�ʱ��ҵϵ�д� Ӣ�żƻ�ͬ����ʱ��Чѵ��ϵ�д�
Ӣ�żƻ�ͬ����ʱ��Чѵ��ϵ�д�
�����Ŀ
 ��2009?����ģ�⣩����ݹ�ҵ��������й�֪ʶ�ش��������⣺
��2009?����ģ�⣩����ݹ�ҵ��������й�֪ʶ�ش��������⣺