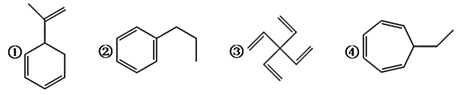
��Ŀ����
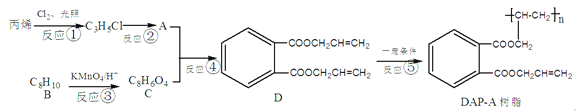
����Ŀ������֧���ķ����廯��������Ҫ�Ļ���ԭ�Ϻͺϳ��м��壬�������ɱ�ϩΪԭ�Ϻϳ���ī����DAP-A��֬�����̡�
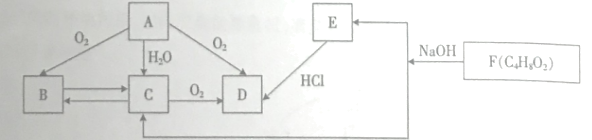
���������գ�
��1��D�ķ���ʽ��____����Ӧ�ܵ�������_____��
��2��д���ṹ��ʽA______��B__________��
��3��д�����з�Ӧ�Ļ�ѧ����ʽ��
��Ӧ�ڣ�________________��
��Ӧ�ܣ�__________________________��
��4����������������C��ͬ���칹�干��___�֡�
����FeCl3��Һ����ɫ�����ܷ���������Ӧ���ۺ���-COOH��
���𰸡�C14H14O4 Ũ����/���� ![]()
![]() CH2=CHCH2Cl+NaOH
CH2=CHCH2Cl+NaOH![]() CH2=CHCH2OH+NaCl
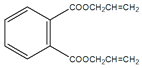
CH2=CHCH2OH+NaCl 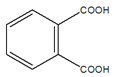 +2CH2=CHCH2OH
+2CH2=CHCH2OH![]()
 +2H2O 10
+2H2O 10
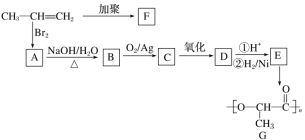
��������
(1)����D�Ľṹ��ʽ��֪��D�ķ���ʽΪC14H14O4����A+C��D��D�Ľṹ��ʽ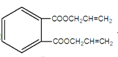 ��֪��Ӧ��Ϊ������Ӧ����Ӧ��Ϊ�Ӿ۷�Ӧ����Dˮ���֪A�Ľṹ��ʽΪCH2=CHCH2OH��C3H5Cl�Ľṹ��ʽΪCH2=CHCH2Cl��C�Ľṹ��ʽΪ
��֪��Ӧ��Ϊ������Ӧ����Ӧ��Ϊ�Ӿ۷�Ӧ����Dˮ���֪A�Ľṹ��ʽΪCH2=CHCH2OH��C3H5Cl�Ľṹ��ʽΪCH2=CHCH2Cl��C�Ľṹ��ʽΪ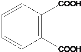 ������B�ķ���ʽ�Ƴ�B�Ľṹ��ʽΪ
������B�ķ���ʽ�Ƴ�B�Ľṹ��ʽΪ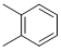 ��
��
(1)����D�Ľṹ��ʽ��֪��D�ķ���ʽΪC14H14O4����Ӧ��Ϊ������Ӧ���������ǣ�Ũ����/���ȣ�
(2)���ݷ�����֪A�Ľṹ��ʽΪCH2=CHCH2OH��B�Ľṹ��ʽΪ ��
��
(3)��Ӧ��Ϊһ�ȱ�ϩ��ˮ�⣺CH2=CHCH2Cl+NaOH![]() CH2=CHCH2OH+NaCl
CH2=CHCH2OH+NaCl
��Ӧ��Ϊ������Ӧ��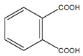 +2CH2=CHCH2OH
+2CH2=CHCH2OH![]()
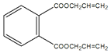 +2H2O��
+2H2O��
(4)����FeCl3��Һ����ɫ��˵�����з��ǻ������ܷ���������Ӧ��˵������ȩ�����ۺ���-COOH����C�Ľṹ��ʽΪ ����������������C��ͬ���칹����10�֣��ṹ��ʽ�ֱ����£�
����������������C��ͬ���칹����10�֣��ṹ��ʽ�ֱ����£�
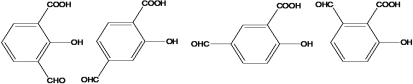
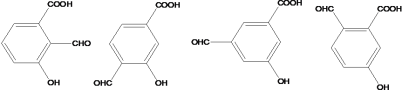
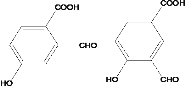 ��
��

����Ŀ��������״̬��,Na��KCI���ڿ��淴Ӧ:Na+KCl![]() NaCl+K,ͨ�������¶�,�����ý���Na����ȡK��
NaCl+K,ͨ�������¶�,�����ý���Na����ȡK��
���� | K | Na | KCl | NaCl |
�۵�/�� | 63.6 | 97.8 | 770 | 801 |
�е�/�� | 774 | 883 | 100 | 1413 |
�����ϱ��и����ʵ��۵�ͷе㣬�Ʋ���Na��KCl��Ӧ��ȡK�ĺ����¶�Ϊ
A.770��B.800 ��C.850 ��D.900 ��