��Ŀ����
����Ŀ����CO2��H2Ϊԭ����ȡ�Ҵ��ķ�ӦΪ 2CO2(g)+6H2(g)CH3CH2OH(g)+3H2O(g)����H<0��ijѹǿ�� ���ܱ�������,��CO2��H2�����ʵ�����Ϊ 1��3 Ͷ��,��ͬ�¶���,�ﵽƽ���ƽ����ϵ�и����ʵ����ʵ�������(y%)���¶ȱ仯��ͼ��ʾ������˵����ȷ����
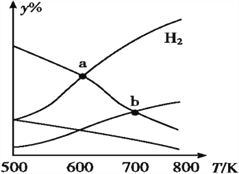
A. a���ƽ�ⳣ��С��b�� B. b��:v��(CO2)=v��(H2O)
C. a��:n(H2)=n(H2O) D. �¶Ȳ���,�������H2,v(CO2)ʼ�ղ���
���𰸡�C
�����������������A����ͼ���֪���¶�Խ�������ĺ���Խ��˵����Ӧ���淴Ӧ�����ƶ���������Ӧ�����Ƿ��ȣ��¶����ߣ�ƽ��Ũ�ȼ�С����ƽ�ⳣ��a>b��A����B��b��ֻ��˵�Ǹ��¶��£�CO2��H2O��Ũ����ͬ��b��ʱ��Ӧ����Ȼ��С����������Ȼ����˵��ƽ��������Ӧ������У�����v����CO2����v�棨H2O����B����C������ͼ�������a��ΪH2��H2O���ʵ����Ľ��㣬������ȣ�C��ȷ��D�����������㶨���������H2������Ӧ���Ũ��������Ӧ������������v��CO2��Ҳ����D����ѡC��

����Ŀ����ǹ�ҵ���Ʊ�Na2S2O3�ķ���֮һ���䷴Ӧԭ��Ϊ��2Na2S+Na2CO3+4SO2==3Na2S2O3+CO2���÷�Ӧ��H>0����ij�о�С����ʵ��������Ʊ�Na2S2O3��5H2O���������¡�

��1������װ����ͼ��ʾ��

��װ��B�������Ǽ���װ��A��SO2������Ч�ʣ�B���Լ��� ____________ ������SO2����Ч�ʵ͵�ʵ��������B����Һ _________________��
��Ϊ��ʹSO2������������ȫ���ڲ��ı�A����ҺŨ�ȡ�����������£����˼�ʱ���跴Ӧ���⣬���ɲ�ȡ�ĺ�����ʩ�� __________________ ������һ����
��2�����豾ʵ�����õ�Na2CO3������NaCl��NaOH�����ʵ�鷽�����м��顣������ʱCaCO3������Һ��pH��10.2����
��� | ʵ����� | Ԥ������ | ���� |
�� | ȡ������Ʒ���Թ��У�������������ˮ��������ܽ⣬ _________�� | �а�ɫ�������� | ��Ʒ��NaCl |
�� | ��ȡ������Ʒ���ձ��У�������������ˮ����ֽ����ܽ⣬________�� | �а�ɫ�������ɣ��ϲ���ҺpH>10.2 | ��Ʒ��NaOH |
��3��Na2S2O3��Һ�Ƕ���ʵ���еij����Լ����ⶨ��Ũ�ȵĹ������£�
��һ����ȷ��ȡa g KIO3����Է�������Ϊ214�����������Һ��
�ڶ������������KI�����H2SO4��Һ���μ�ָʾ����
�������� ��Na2S2O3��Һ�ζ����յ㣬����Na2S2O3��Һ�����ΪV mL��
��c(Na2S2O3)��_________mol��L��1��
��֪��IO3-+5I-+6H+= 3I2��3H2O ��2S2O32����I2=S4O62����2I��
��4��ijͬѧ��һ���͵ڶ����IJ������ܹ淶������������̫����������õ�Na2S2O3Ũ�ȿ���__________����������Ӱ��������ƫ��������ƫ��������ԭ����_________________________________���������ӷ���ʽ��ʾ����
����Ŀ��һ�������£����и���������һ��ʵ��ͼ����ʾת����ϵ����

ѡ�� | X | Y | Z | W |
A | Al | Al2O3 | NaAlO2 | Al(OH)3 |
B | Fe3O4 | Fe | FeCl2 | FeCl3 |
C | H2SO4 | SO2 | S | SO3 |
D | CH3CH2Br | CH2=CH2 | C2H5OH | CH2BrCH2Br |
A. A B. B C. C D. D



