��Ŀ����
��12�֣���ҵ������������Ҫ�ɷ���Al2O3������������Fe2O3��SiO2����ȡұ������ԭ����������������������ͼ��
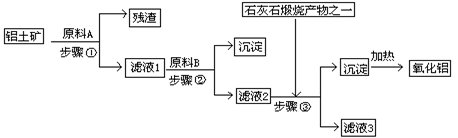
(1) ԭ��A��������____������ٷ�Ӧ�����ӷ���ʽ��_________________��
(2) ��Һ1��Ҫ�����Թ���ԭ��B��ԭ��B�Ļ�ѧʽ��__������ڷ�Ӧ�����ӷ���ʽ��
(3) ����۵Ļ�ѧ����ʽ�ǣ�_____________________________________________��
(4) ���ʡȥ����٣����ܽ��������ǴӼ���ԭ��B��ʼ�������������䣬��������������ȡ��ʲôӰ��__________________________________________________��

(1) ԭ��A��������____������ٷ�Ӧ�����ӷ���ʽ��_________________��
(2) ��Һ1��Ҫ�����Թ���ԭ��B��ԭ��B�Ļ�ѧʽ��__������ڷ�Ӧ�����ӷ���ʽ��
(3) ����۵Ļ�ѧ����ʽ�ǣ�_____________________________________________��
(4) ���ʡȥ����٣����ܽ��������ǴӼ���ԭ��B��ʼ�������������䣬��������������ȡ��ʲôӰ��__________________________________________________��

��

��ϰ��ϵ�д�
�����Ŀ
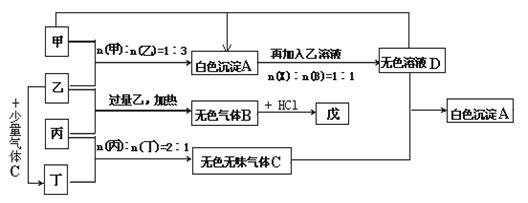
 B���������ǵ��ʣ�д���������Һ���ܷ���ʽ�� ��
B���������ǵ��ʣ�д���������Һ���ܷ���ʽ�� ��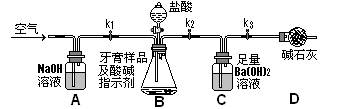
 2MgO����3Mg+N2
2MgO����3Mg+N2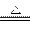 MgO+H2�� ��Mg3N2 +6H2O ===3Mg(OH)2+2NH3��
MgO+H2�� ��Mg3N2 +6H2O ===3Mg(OH)2+2NH3��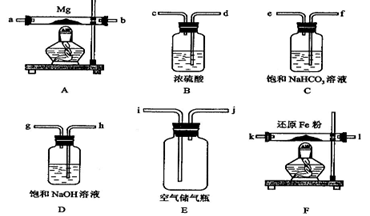
 ��
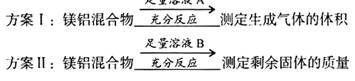
�� �������Ӳ������ܼ���AlCI3����ʧ����ش��������⣺
�������Ӳ������ܼ���AlCI3����ʧ����ش��������⣺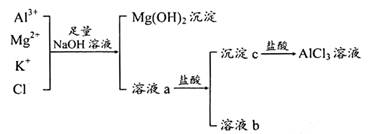
 ����ת�Ƶ�����ƿ�С�
����ת�Ƶ�����ƿ�С� ��������ƿ���ӣ���ҡ�ȡ�
��������ƿ���ӣ���ҡ�ȡ�