��Ŀ����
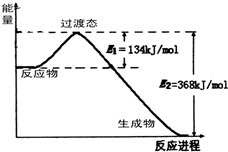
��֪�Ȼ�ѧ����ʽ2SO2(g) + O2(g)  2 SO3(g) ��H = ��Q kJ/mol��Q��0����������˵��
2 SO3(g) ��H = ��Q kJ/mol��Q��0����������˵��
��ȷ���ǣ� ��.
| A��2 mol SO2(g)��1 mol O2(g)�����������2 mol SO3(g)��������� |
| B����2 mol SO2(g)��1 mol O2(g)����һ�ܱ������г�ַ�Ӧ��ų�QkJ������ |
| C�������¶ȣ�ƽ��������Ӧ�����ƶ��������Ȼ�ѧ����ʽ�е�Qֵ��С |
| D���罫һ����SO2(g)��O2(g)����ij�ܱ������г�ַ�Ӧ�����QkJ����˹�������2 mol SO2(g)������ |
D
����

��ϰ��ϵ�д�
�����Ŀ
 ��Դ�ǹ��÷�չ����Ҫ�������ҹ�Ŀǰʹ�õ���Դ��Ҫ�ǻ�ʯȼ�ϣ�
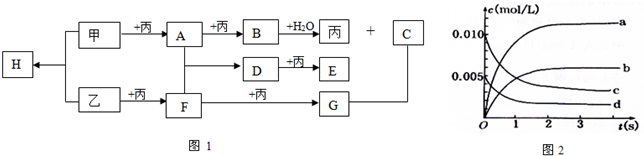
��Դ�ǹ��÷�չ����Ҫ�������ҹ�Ŀǰʹ�õ���Դ��Ҫ�ǻ�ʯȼ�ϣ�  ����A��B��C����ͼ1ת����ϵ��
����A��B��C����ͼ1ת����ϵ��