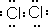��Ŀ����
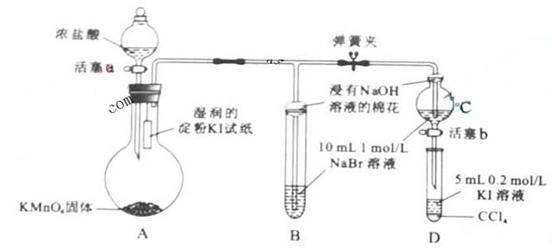
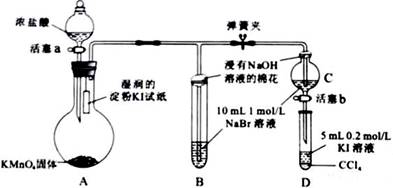
 Ϊ��֤±�ص��������Ե����ǿ����ijС������ͼ��ʾװ�ý������飨�г���������ȥ���������Ѽ��飩��
Ϊ��֤±�ص��������Ե����ǿ����ijС������ͼ��ʾװ�ý������飨�г���������ȥ���������Ѽ��飩��
������̣�
��.���ɼУ�����a���μ�Ũ���ᡣ
��.��B��C�е���Һ����Ϊ��ɫʱ���н����ɼС�
��.��B����Һ�ɻ�ɫ��Ϊ��ɫʱ���رջ���a��
��������
��1��A�в�������ɫ���壬�����ʽ�� ��
��2����֤������������ǿ�ڵ��ʵ�������� ��
��3��B����Һ������Ӧ�����ӷ���ʽ�� ��
��4��Ϊ��֤���������ǿ�ڵ⣬���̢��IJ����������� ��
��5�����̢�ʵ���Ŀ���� ��
��6���ȡ��塢�ⵥ�ʵ�������������ԭ��ͬ����Ԫ�ش��ϵ��� ���õ�������������
������������Լ��ǣ�1��~��3���ͣ�6���ʣ����ڻ���֪ʶ��ֱ�ӿ��顣ѧ��ֻҪ�жϳ�����ɫ������������ǰ��������Ȼ˳�����µó��𰸡��Ƚ��ѵ��£�4����5�����ʡ�
��֤���������ǿ�ڵ⣬��Ҫ��װ��D����ɣ���ʱC���������ɡ���ɸ÷�Ӧ�������b��������C����Һ����D�У�Ȼ��رջ���b��ȡ��D����ֹ��CCl4����Һ��Ϊ�Ϻ�ɫ��ͨ���е�������֤�����������ǿ�ڵ⡣����ѧ��Ӧ��ʧ���ڱ�����ȷ����λ����ȱ�ٲ������裬�������رջ�������5������Ҫѧ����ȷʵ�鲽�������ã���ʵ����ȷ��C�Ļ�ɫ��Һ����Cl2���ų�Cl2�����û���ʵ��ĸ��š�

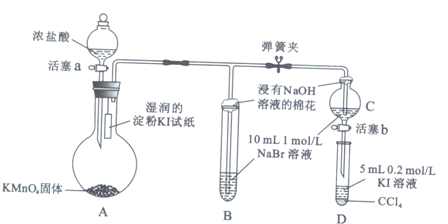
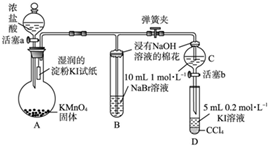 Ϊ��֤±�ص��������Ե����ǿ����ijС������ͼ��ʾװ�ý���ʵ�飨�г���������ȥ���������Ѽ��飩
Ϊ��֤±�ص��������Ե����ǿ����ijС������ͼ��ʾװ�ý���ʵ�飨�г���������ȥ���������Ѽ��飩