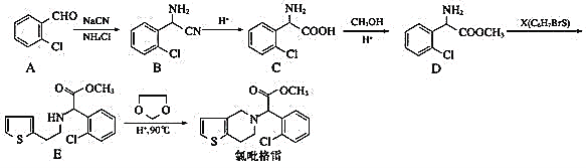
��Ŀ����
����Ŀ��Ϊ�ⶨij�л�������A�Ľṹ����������ʵ�飺
��1����һ�������л���A���������г��ȼ�գ�ʵ���ã�����5.4g H2O��8.8g CO2����������6.72L����״���£�������л����ʵ��ʽ��__��
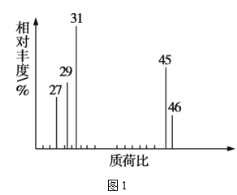
��2���������Dzⶨ���л����������Է����������õ���ͼ1��ʾ������ͼ��������Է�������Ϊ___�������ʵķ���ʽ��__��
��3�����ݼۼ����ۣ�Ԥ��A�Ŀ��ܽṹ��д���ṹ��ʽ��___��
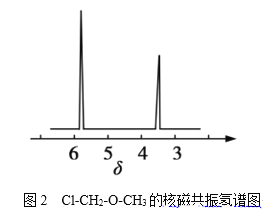
��4���˴Ź��������ܶ��л�������в�ͬλ�õ���ԭ�Ӹ�����ͬ�����շ壨�źţ����������շ壨�źţ�����ȷ����������ԭ�ӵ��������Ŀ�������ȼ����ѣ�Cl-CH2-O-CH3����2����ԭ�ӣ��ĺ˴Ź���������ͼ2��ʾ��
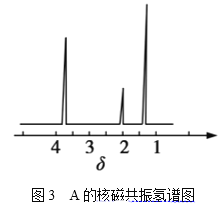
���ⶨ���л���A�ĺ˴Ź�������ͼ��ͼ3��ʾ����A�Ľṹ��ʽΪ__��
���𰸡�C2H6O 46 C2H6O CH3CH2OH��CH3-O-CH3 CH3CH2OH
��������
Ũ��������Ϊˮ����������ʯ������Ϊ������̼���������Դ˿�ȷ���л�����C��Hԭ�Ӹ�����ֵ��������������������ȷ���л����и�ԭ�Ӹ�����ֵ�������л���ԭ�Ӹ�����ֵ��ȷ�����ʽ�������Է���������ȷ���л������ʽ�������л������ʽ��ϼۼ����ۿ�ȷ���л���Ŀ��ܽṹ���ٸ�������ͼ��ȷ���л���Ľṹʽ���Դ˷�����
(1)����5.4g H2O��8.8g CO2��H2O�����ʵ���Ϊ![]() ����0.6mol����ԭ�ӣ�CO2���ʵ���Ϊ
����0.6mol����ԭ�ӣ�CO2���ʵ���Ϊ![]() ����̼�����ʵ���Ϊ0.2mol��O2�����ʵ���Ϊ
����̼�����ʵ���Ϊ0.2mol��O2�����ʵ���Ϊ![]() ������ԭ�ӵ����ʵ���Ϊ0.6mol�����ݷ���ʽ����ԭ���غ�ã��л����е���ԭ�����ʵ���Ϊ
������ԭ�ӵ����ʵ���Ϊ0.6mol�����ݷ���ʽ����ԭ���غ�ã��л����е���ԭ�����ʵ���Ϊ![]() �����л���C��H��O�����ʵ���֮��Ϊ2��6��1��ʵ��ʽΪC2H6O��
�����л���C��H��O�����ʵ���֮��Ϊ2��6��1��ʵ��ʽΪC2H6O��
(2)������ͼ�ɵ���Է�������Ϊ46�������ʽΪC2H6O��
(3)��A����ʽC2H6O��֪AΪ���ͻ�����Ʋ���ṹ��ʽΪCH3CH2OH��CH3-O-CH3��
(4)����A�ĺ˴Ź�������ͼ��֪A��3�ֲ�ͬ������Hԭ�ӣ�CH3-O-CH3ֻ��1����ԭ�ӣ�CH3CH2OH��3�ֲ�ͬ������Hԭ�ӣ���AΪCH3CH2OH��

 Ӧ������ҵ��ϵ�д�
Ӧ������ҵ��ϵ�д�