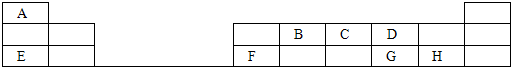
��Ŀ����
��A��D��E��G����Ԫ����ɵĻ�����ķ���ʽΪ![]() ����֪��
����֪��
(1)![]() ��������
��������![]() ��
��
(2)ȡ8.74g�û���������ˮ���������ǿ���������������е�E��GԪ����ȫת������̬������![]() 2.688L(��״����)�����ܶ�Ϊ1.965g/L��
2.688L(��״����)�����ܶ�Ϊ1.965g/L��![]() ��ʹ����ʯ��ˮ����ǣ�
��ʹ����ʯ��ˮ����ǣ�
(3)����������Ӧ��A��D�������Ӵ�������Һ�У�����Һ�������������ۣ�����ʹ����ȫ����ԭ�����ĵ���������Ϊ0.56g��
(4)��������DԪ�ص���������Ϊ12.8����
��ͨ�����������ȷ��E��G��D��A����ʲôԪ�أ����![]() ��ֵ����д���û�����Ļ�ѧʽ��
��ֵ����д���û�����Ļ�ѧʽ��
������
|
�𰸣� (1)E��̼��G������ (2) (3)A�Ǽأ��û����ﻯѧʽΪ ����ʵ�����ݣ�ͨ�����㣬������������ȷ��δ֪��Ļ�ѧʽ���Ƕ����о���ѧ��һ����Ҫ���⣮�ڱ����У�
�ִ� ������̼�����ʵ����ǣ� D�����Ǿ��������ԵĽ������ӣ��ù������ۻ�ԭD���ӣ�Fe������Ϊ
��ÿ��D���ӵõ�1�����ӣ���ԭ��D�����ʵ���Ϊ0.02mol���ڸû�������D��(
�� �� DԪ�ص����ԭ������Ϊ��
�ɴ˿�֪D������D������
AԪ�ص����ԭ������Ϊ��
������ͨ�����������δ֪��Ļ�ѧʽΪ (1)E��̼��G������ (2) (3)A�Ǽأ��û����ﻯѧʽΪ |
��ʾ��
|
��Fe�۹��������£�Feֻ�ܱ�����Ϊ |

 ��������һ���þ�ϵ�д�
��������һ���þ�ϵ�д� Сѧ��10����Ӧ����ϵ�д�
Сѧ��10����Ӧ����ϵ�д�