��Ŀ����
������KMnO4����ŨHCl�ķ�Ӧ����ʽ���£�2KMnO4 ��16HCl��Ũ��=2KCl��2MnCl 2��5Cl 2����8H2O
��1���á�˫���ŷ���������Ӧ�е���ת�Ƶķ������Ŀ��
��2������֪ ��������Ϸ�Ӧԭ������MnO2��KMnO4��Cl2����������ǿ������˳���ǣ� ��
��������Ϸ�Ӧԭ������MnO2��KMnO4��Cl2����������ǿ������˳���ǣ� ��
��3������63.2g������زμӷ�Ӧ����������HCl�����ʵ���Ϊ mol��������Cl2�ڱ�״���µ����Ϊ L��
��1���á�˫���ŷ���������Ӧ�е���ת�Ƶķ������Ŀ��
��2������֪
 ��������Ϸ�Ӧԭ������MnO2��KMnO4��Cl2����������ǿ������˳���ǣ� ��
��������Ϸ�Ӧԭ������MnO2��KMnO4��Cl2����������ǿ������˳���ǣ� ����3������63.2g������زμӷ�Ӧ����������HCl�����ʵ���Ϊ mol��������Cl2�ڱ�״���µ����Ϊ L��
��1��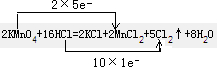 ��2��KMnO4 MnO2 Cl2 ��3��2 22.4
��2��KMnO4 MnO2 Cl2 ��3��2 22.4
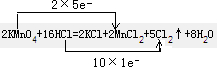 ��2��KMnO4 MnO2 Cl2 ��3��2 22.4
��2��KMnO4 MnO2 Cl2 ��3��2 22.4 �����������1��˫���ŷ������Ӧ�е��ӵ�ʧ��ĿΪ
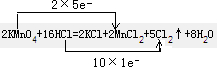 ��
����2��2KMnO4 ��16HCl��Ũ��="2KCl" ��2MnCl 2��5Cl 2�� ��8H2O�˷�Ӧ��ȡCl2����Ҫ���������ڷ�Ӧ
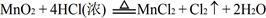 ��Ҫ���ȣ���MnO2���������� Cl2���������������������KMnO4 >MnO2 >Cl2
��Ҫ���ȣ���MnO2���������� Cl2���������������������KMnO4 >MnO2 >Cl2��3��������ص��������������Լ����ĵ�����֮��Ĺ�ϵΪ��2KMnO4��5Cl2��16HCl��������������Ͳμӷ�Ӧ���������֮��Ϊ��10 :16 ��5 :8 ��63.2g ����0.4mol��KMnO4��ȫ��Ӧʱ��������Cl2Ϊ1.0mol���ڱ�״���µ����Ϊ1.0mol��22.4L/mol=22.4L�����ĵ�HClΪ3.2mol����������HClΪ2.0mol.

��ϰ��ϵ�д�
�����Ŀ
 8Cu+4FeO+2Fe2O3+16SO2
8Cu+4FeO+2Fe2O3+16SO2


 ��CO2+��K2SO4+��MnSO4+��H2O
��CO2+��K2SO4+��MnSO4+��H2O
 HNO3��д��OH��NO��Ӧ�Ļ�ѧ����ʽ��_____________���÷�Ӧ�б�������Ԫ���� ��
HNO3��д��OH��NO��Ӧ�Ļ�ѧ����ʽ��_____________���÷�Ӧ�б�������Ԫ���� ��

