ЬтФПФкШн
ЁОЬтФПЁП2SO2(g)ЃЋO2(g)==2SO3(g)ЗДгІЙ§ГЬжаЕФФмСПБфЛЏШчЯТЭМЫљЪОЁЃ
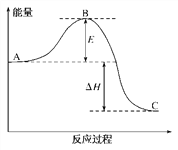
вбжЊ1mol SO2(g)бѕЛЏЮЊ1mol SO3(g)ЕФІЄHЃНЃ99 kJЁЄmolЃ1
ЧыЛиД№ЯТСаЮЪЬтЃК
(1)ЭМжаAЁЂCЗжБ№БэЪО__________ЕФФмСПЁЂ___________ЕФФмСПЃЛ
(2)ЭМжаІЄHЃН______________________ЃЛ
(3)вбжЊЕЅжЪСђЕФШМЩеШШЮЊЃ296 kJЁЄmolЃ1ЃЌМЦЫугЩS(s)ЩњГЩ3mol SO3(g)ІЄH=___________ЃЛ
(4)ЛЏбЇЗДгІЕФьЪБфгыЗДгІЮяКЭЩњГЩЮяЕФМќФмгаЙиЁЃ
вбжЊЃКЂйH2(g)ЃЋCl2(g)=2HCl(g) ІЄHЃНЃ185 kJЁЄmolЃ1
ЂкHЁЊHМќФмЮЊ436kJЁЄmolЃ1ЃЌClЁЊClМќФмЮЊ247kJЁЄmolЃ1ЁЃдђHЁЊClМќФмЮЊ____________________ЁЃ
ЁОД№АИЁП ЗДгІЮя ЩњГЩЮя Ѓ198 kJЁЄmolЃ1 ІЄHЃНЃ1185 kJЁЄmolЃ1 434 kJЁЄmolЃ1
ЁОНтЮіЁПЃЈ1ЃЉЭМжаAЁЂCЗжБ№БэЪОЗДгІЮязмФмСПЁЂЩњГЩЮязмФмСПЃЌEЮЊЛюЛЏФмЃЌЙЪД№АИЮЊЃКЗДгІЮяЃЛЩњГЩЮяЃЛ
ЃЈ2ЃЉвђ1mol SO2ЃЈgЃЉбѕЛЏЮЊ1mol SO3ЕФЁїH=-99kJmol-1ЃЌЫљвд2molSO2ЃЈgЃЉбѕЛЏЮЊ2molSO3ЕФЁїH=-198kJmol-1ЃЌдђ2SO2ЃЈgЃЉ+O2ЃЈgЃЉ=2SO3ЃЈgЃЉЁїH=-198KJmol-1ЃЌЙЪД№АИЮЊЃК-198 kJЁЄmolЃ1ЃЛ
ЃЈ3ЃЉЕЅжЪСђЕФШМЩеШШЮЊ296kJmol-1ЃЌдђSЃЈsЃЉ+O2ЃЈgЃЉ=SO2ЃЈgЃЉЁїH=-296kJmol-1ЃЌЖј1molSO2ЃЈgЃЉбѕЛЏЮЊ1molSO3ЕФЁїH=-99kJmol-1ЃЌдђSO2ЃЈgЃЉ+![]() O2ЃЈgЃЉ=SO3ЃЈgЃЉЁїH=-99kJmol-1ЃЌгЩИЧЫЙЖЈТЩПЩЕУЃКSЃЈsЃЉ+
O2ЃЈgЃЉ=SO3ЃЈgЃЉЁїH=-99kJmol-1ЃЌгЩИЧЫЙЖЈТЩПЩЕУЃКSЃЈsЃЉ+![]() O2ЃЈgЃЉ=SO3ЃЈgЃЉЁїH=-296kJmol-1+ЃЈ-99kJmol-1ЃЉ=-395kJmol-1ЃЌдђ3SЃЈsЃЉ+
O2ЃЈgЃЉ=SO3ЃЈgЃЉЁїH=-296kJmol-1+ЃЈ-99kJmol-1ЃЉ=-395kJmol-1ЃЌдђ3SЃЈsЃЉ+![]() O2ЃЈgЃЉЈT3SO3ЃЈgЃЉЁїH=Ѓ1185 kJmol-1ЃЌЙЪД№АИЮЊЃКЃ1185 kJmol-1ЃЛ
O2ЃЈgЃЉЈT3SO3ЃЈgЃЉЁїH=Ѓ1185 kJmol-1ЃЌЙЪД№АИЮЊЃКЃ1185 kJmol-1ЃЛ
(4)ЂйH2ЃЈgЃЉ+Cl2ЃЈgЃЉЈT2HClЃЈgЃЉЁїH=-185kJmol-1ЃЌЂкЁїH298ЃЈH2ЃЉ=+436kJmol-1ЃЌЂлЁїH298ЃЈCl2ЃЉ=+247kJmol-1ЃЌдђЁїH=ЁїH298ЃЈH2ЃЉ+ЁїH298ЃЈCl2ЃЉ-2ЁСЁїH298ЃЈHClЃЉ=-185kJmol-1ЃЌЁїH298ЃЈHClЃЉ=![]() ЃЈЁїH298ЃЈH2ЃЉ+ЁїH298ЃЈCl2ЃЉ+185KJ/molЃЉ=+434KJ/molЃЌЙЪД№АИЮЊЃК+434KJ/mol
ЃЈЁїH298ЃЈH2ЃЉ+ЁїH298ЃЈCl2ЃЉ+185KJ/molЃЉ=+434KJ/molЃЌЙЪД№АИЮЊЃК+434KJ/mol

 гЂВХМЦЛЎЦкФЉЕїбаЯЕСаД№АИ
гЂВХМЦЛЎЦкФЉЕїбаЯЕСаД№АИ