��Ŀ����
����Ŀ������ѧ����ѡ��2��ѧ�뼼������п��ZnS����һ����Ҫ�Ļ���ԭ�ϣ�������ˮ��������п�ķ���п����ȡ���乤����������ͼ��ʾ��

��1��Ϊ���п�ҵĽ�ȡ�ʣ��ɲ��õķ����� �����������
����ĥ ����ν�ȡ �������¶� ����ѹ ������
��2�����������������е������� ��д��ѧʽ����
��3���������пɵ�Cd���ʣ�Ϊ���������µ����ʣ��Լ�bӦΪ____________��
��4�������������Ի���Na2SO4����ȡNa2S��
������ZnS�����Ƿ�ϴ�Ӹɾ��ķ����� ��

��Na2S���ɵ����ʵ�����Na2SO4��CH4�ڸ��¡�������������ȡ����ѧ��Ӧ����ʽΪ__________��
����֪Na2SO4��10H2O��Na2SO4���ܽ�����¶ȱ仯������ͼ������Һ�еõ�Na2SO4��10H2O�IJ��������� ��
��5���������������ZnCO3Ϊb mol������������CdΪd mol�����õ�VL�����ʵ���Ũ��Ϊc mol/L��Na2SO4��Һ��������������п���к���пԪ�ص�����Ϊ____________��
���𰸡���1���٢ڢۢ�
��2�� Fe��OH��3��ZnCO3 �� ֻдFe��OH��3���۷���
��3��Zn�����
��4����ȡ����ϴ��Һ�������Թܣ��μӼ���BaCl2��Һ�������ֻ�����δϴ������֮����ϴ��
��Na2SO4��CH4![]() =Na2S��2H2O��CO2
=Na2S��2H2O��CO2
������Ũ�������½ᾧ������
��5��65��Vc��b��d��g
��������
�����������1����ĥ����ν�ȡ�������¶ȡ����趼�����п�ҵĽ�ȡ�ʣ���ѡ�٢ڢۢ���
��2��п�ҽ�ȡҺ�м���H2O2��Fe2+������ΪFe3+����̼��п��pH��Fe��OH��3���������������������е�������Fe��OH��3��ZnCO3��
��3����Һ���к�Zn2+��Cd2+����������Ϊ���������µ����ʣ��Լ�bӦΪZn��
��4����ͨ������Na2SO4����ȡNa2S�������ZnS�����Ƿ�ϴ�Ӹɾ��ķ�����ȡ����ϴ��Һ�������Թܣ��μӼ���BaCl2��Һ�������ֻ�����δϴ������֮����ϴ����
�������ʵ�����Na2SO4��CH4�ڸ��¡�������������ȡNa2S����ѧ��Ӧ����ʽΪNa2SO4��CH4![]() =Na2S��2H2O��CO2��
=Na2S��2H2O��CO2��
�����ܽ�����߿�֪������Һ�еõ�Na2SO4��10H2O�IJ�������������Ũ�������½ᾧ�����ˣ�
��5������������CdΪd mol���������û�Cd��Zn��d mol�����õ�VL�����ʵ���Ũ��Ϊc mol/L��Na2SO4��Һ����ZnSO4���ʵ���ΪcVmol������пԪ���غ��п���к���пԪ�ص�����Ϊ65��cV-b-d��g��

����Ŀ��[��ѧ����ѡ��3�����ʽṹ������]����Ge���ǵ��͵İ뵼��Ԫ�أ��ڵ��ӡ����ϵ�����Ӧ�ù㷺���ش��������⣺
��1����̬Geԭ�ӵĺ�������Ų�ʽΪ[Ar]____________����__________��δ�ɶԵ��ӡ�
��2��Ge��C��ͬ��Ԫ�أ�Cԭ��֮������γ�˫������������Geԭ��֮�������γ�˫������������ԭ�ӽṹ�Ƕȷ�����ԭ����________________��
��3���Ƚ�������±������۵�ͷе㣬������仯���ɼ�ԭ��_____________________��
GeCl4 | GeBr4 | GeI4 | |
�۵�/�� | 49.5 | 26 | 146 |
�е�/�� | 83.1 | 186 | Լ400 |
��4�������ԭCO2�Ʊ�CH4��Ӧ�У���״����Zn2GeO4�Ǹ÷�Ӧ�����ô�����Zn��Ge��O�縺���ɴ���С��˳����______________��
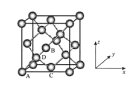
��5��Ge�������н��ʯ�ͽṹ������Geԭ�ӵ��ӻ���ʽΪ_______________________����֮����ڵ���������_____________��
��6����������������Ҫ�أ�
��ԭ�������������ʾ�����ڲ���ԭ�ӵ����λ�ã���ͼΪGe�����ľ���������ԭ���������AΪ��0,0,0����BΪ��![]() ��0��
��0��![]() ����C��
����C��![]() ��
��![]() ��0������Dԭ�ӵ��������Ϊ______��
��0������Dԭ�ӵ��������Ϊ______��
���������������������Ĵ�С����״����֪Ge�����ľ�������a=565.76 pm�����ܶ�Ϊ__________g��cm-3���г�����ʽ��������
����Ŀ��ƻ����(ACV)��һ����ƻ�����Ͷ��ɵ�������Ʒ�����нⶾ����֬��ҩЧ��ƻ������һ�ֳ������л��ᣬ��ṹ��ʽΪ![]() ��
��
��1��ƻ�����к��еĹ����ŵ�������________��_____________��
��2��ƻ���ķ���ʽΪ__________________��
��3��1 molƻ���������������Ʒ�Ӧ�������ɱ�״���µ�����________L��
��4��ƻ�����ܷ����ķ�Ӧ��________��
A����NaOH��Һ��Ӧ |
B����ʯ����Һ���� |
C����������һ������������ |
D�����Ҵ���һ������������ |