��Ŀ����
��1����NaHCO3��Һ��Al2(SO4)3��Һ��ϣ���ط�Ӧ�����ӷ���ʽ��____________��
��2����Ũ�Ⱦ�Ϊ0.2 mol��L-1��Na2CO3��Һ��NaHCO3��Һ���Իش��������⣺
�������ࣺNa2CO3__________NaHCO3��Һ�����������������������
������Һ�ֱ����0.1 molNaOH���壬�ָ������£�Na2CO3��Һ�е�c(CO32-)____________�����������С�����䡱����NaHCO3��Һ�е�c(HCO3-)_________�����������С�����䡱����
��3�������£���Ϊ0.1 mol��L-1��HA��Һ��pH��1����Ϊ0.1 mol��L-1��BOH��Һ������Һ��c(OH-):c(H+)=1012����ֱ�д���ס�������Һ�����ʵĵ��뷽��ʽ��
��__________________����_________________��
��4����ͬ�����£�����Ũ�Ⱦ�Ϊ0.1 mol��L-1��NaCl��Һ��CH3COONa��Һ����Һ�е�����������NaCl��Һ__________CH3COONa��Һ�����������������������
��5�����ʵ���Ũ����ͬ�� �ٰ�ˮ ���Ȼ�� ��̼����� ��������� �������������Һ��c(NH4+)�ɴ�С��˳��Ϊ__________________��
 ����������ϵ�д�
����������ϵ�д�

 4NO(g)+CO2(g)+2H2O(g) ��H=-574kJ/mol
4NO(g)+CO2(g)+2H2O(g) ��H=-574kJ/mol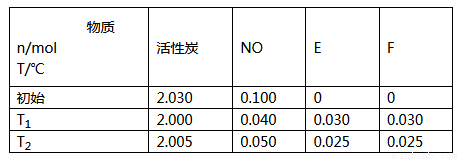
 2CO2+N2��ij�¶�ʱ����1L�ܱ������г���0.1molCO��0.1mol NO��5sʱ��Ӧ�ﵽƽ�⣬���NO��Ũ��Ϊ0.02mol/L����Ӧ��ʼ��ƽ��ʱ��NO��ƽ����Ӧ����v(NO)=________�������¶��£�ijʱ�̲��CO��NO��N2��CO2��Ũ�ȷֱ�Ϊ0.01mol/L��amol/L��0.01mol/L��0.04mol/L��Ҫʹ��Ӧ������Ӧ������У�a��ȡֵ��ΧΪ_____________��
2CO2+N2��ij�¶�ʱ����1L�ܱ������г���0.1molCO��0.1mol NO��5sʱ��Ӧ�ﵽƽ�⣬���NO��Ũ��Ϊ0.02mol/L����Ӧ��ʼ��ƽ��ʱ��NO��ƽ����Ӧ����v(NO)=________�������¶��£�ijʱ�̲��CO��NO��N2��CO2��Ũ�ȷֱ�Ϊ0.01mol/L��amol/L��0.01mol/L��0.04mol/L��Ҫʹ��Ӧ������Ӧ������У�a��ȡֵ��ΧΪ_____________��
 Si3N4��12HCl���йظ÷�Ӧ˵����ȷ����
Si3N4��12HCl���йظ÷�Ӧ˵����ȷ����