��Ŀ����
�����ű�����ȫ����̡�ʡ��֮�Ƶĸ�����������ȴƵƵ�ܵ���������ţ�������ˮԴ��ˮ��״�����棬����9�������е�34������ʽ��������ˮԴ��ˮ�ʴ����Ϊ73.3%��������ͬ�������½������Ա������������ǿ̲��ݻ������Σ�
�����������ǿ��ƶ���������Ⱦ����Ҫ�����ֶΣ�
��1�����ú�ˮ������һ����Ч�ķ������乤��������ͼ1��ʾ��
ij�о�С��Ϊ̽����ߺ���������SO2������Ч�ʵĴ�ʩ����������Ȼ��ˮ��pH��8�����պ���������ģ��ʵ�飬ʵ������ͼ2��ʾ��
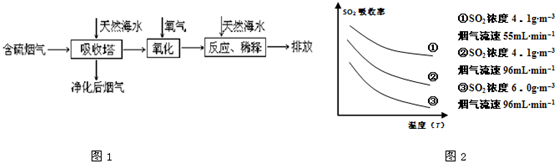
�ٸ���ͼʾʵ������Ϊ�����һ��Ũ�Ⱥ���������SO2������Ч�ʣ����д�ʩ��ȷ����
A������ͨ�뺬���������¶� B����Сͨ�뺬������������
C��������Ȼ��ˮ�Ľ����� D������Ȼ��ˮ�м�����ʯ��
����Ȼ��ˮ�����˺��������������H2SO3��ʹ�ÿ����е���������������д���÷�Ӧ�Ļ�ѧ����ʽ��
��2��ʯ��ʯ-ʯ��ʪ�����������ռ����Ĺ���ԭ���������еĶ��������뽬Һ�е�̼����Լ�����Ŀ�����Ӧ����ʯ�ࣨCaSO4?2H2O����д���÷�Ӧ�Ļ�ѧ����ʽ��
���ؽ������ӶԺ������������������Ⱦ��ij��������ˮ��pH��2���к���Ag+��Pb2+���ؽ������ӣ���Ũ�ȸ�ԼΪ0.01mol?L-1���ŷ�ǰ���ó�������ȥ���������ӣ������й��������£�
��3������Ϊ����ˮ��Ͷ��
A��NaOH B��Na2S C��KI D��Ca��OH��2
��4�����ֻ����ʯ�Ҵ�������Pb2+�ķ�ˮ��ʹ��Һ��pH=8.0��������ķ�ˮ�У�c��Pb2+��=
�����������ǿ��ƶ���������Ⱦ����Ҫ�����ֶΣ�
��1�����ú�ˮ������һ����Ч�ķ������乤��������ͼ1��ʾ��
ij�о�С��Ϊ̽����ߺ���������SO2������Ч�ʵĴ�ʩ����������Ȼ��ˮ��pH��8�����պ���������ģ��ʵ�飬ʵ������ͼ2��ʾ��

�ٸ���ͼʾʵ������Ϊ�����һ��Ũ�Ⱥ���������SO2������Ч�ʣ����д�ʩ��ȷ����
AB
AB
��������ĸ��ţ�A������ͨ�뺬���������¶� B����Сͨ�뺬������������
C��������Ȼ��ˮ�Ľ����� D������Ȼ��ˮ�м�����ʯ��
����Ȼ��ˮ�����˺��������������H2SO3��ʹ�ÿ����е���������������д���÷�Ӧ�Ļ�ѧ����ʽ��
2H2SO3+O2=2H2SO4
2H2SO3+O2=2H2SO4
��������ġ���ˮ����Ҫ�ô�������Ȼ��ˮ��֮��Ϻ�����ŷţ��ò�������ҪĿ�����к͡�ϡ�;�����������ˮ�����ɵ��ᣨH+��
�к͡�ϡ�;�����������ˮ�����ɵ��ᣨH+��
����2��ʯ��ʯ-ʯ��ʪ�����������ռ����Ĺ���ԭ���������еĶ��������뽬Һ�е�̼����Լ�����Ŀ�����Ӧ����ʯ�ࣨCaSO4?2H2O����д���÷�Ӧ�Ļ�ѧ����ʽ��
2CaCO3+2SO2+O2+4H2O�T2CaSO4?2H2O+2CO2
2CaCO3+2SO2+O2+4H2O�T2CaSO4?2H2O+2CO2
�����ؽ������ӶԺ������������������Ⱦ��ij��������ˮ��pH��2���к���Ag+��Pb2+���ؽ������ӣ���Ũ�ȸ�ԼΪ0.01mol?L-1���ŷ�ǰ���ó�������ȥ���������ӣ������й��������£�
| ���ܵ���� | AgI | AgOH | Ag2S | PbI2 | Pb��OH��2 | PbS |
| Ksp | 8.3��10-17 | 5.6��10-8 | 6.3��10-50 | 7.1��10-9 | 1.2��10-15 | 3.4��10-28 |
B
B
������ĸ��ţ�������Ч����ã�A��NaOH B��Na2S C��KI D��Ca��OH��2
��4�����ֻ����ʯ�Ҵ�������Pb2+�ķ�ˮ��ʹ��Һ��pH=8.0��������ķ�ˮ�У�c��Pb2+��=
1.2��10-3mol?L-1
1.2��10-3mol?L-1
��������Ҫ����ˮ�ۺ��ŷű�Ϊc��Pb2+������1.0��l0-8mol?L-1���ʴ�����ķ�ˮ�Ƿ�����ŷű�����
��
����ǡ�������������1���ٸ���ͼ2��֪�¶ȡ����ٶ�����SO2������Ч�ʣ�
�ڸ���ʹ�ÿ����е�������H2SO3������������������Ӧ������ϵ�Ŀ�ģ�
��2���ɶ��������뽬Һ�е�̼����Լ�����Ŀ�����Ӧ����ʯ������д��Ӧ����ʽ��
��3���ɱ����е����ݿ�֪���ܶȻ�ԽС��Խ��ת��Ϊ������
��4������Pb��OH��2���ܶȻ����м��㣬�����ŷű��Ա���˵���Ƿ�����ŷű���
�ڸ���ʹ�ÿ����е�������H2SO3������������������Ӧ������ϵ�Ŀ�ģ�
��2���ɶ��������뽬Һ�е�̼����Լ�����Ŀ�����Ӧ����ʯ������д��Ӧ����ʽ��
��3���ɱ����е����ݿ�֪���ܶȻ�ԽС��Խ��ת��Ϊ������
��4������Pb��OH��2���ܶȻ����м��㣬�����ŷű��Ա���˵���Ƿ�����ŷű���
����⣺��1������ͼ2��֪����pHһ����ǰ���£��¶�Խ�͡�����ԽС������SO2������Ч��Խ�ߣ��ʴ�Ϊ��AB��
���ɿ����е�������H2SO3����Ϊ���ᣬ�÷�ӦΪ2H2SO3+O2=2H2SO4��������ġ���ˮ����Ҫ�ô�������Ȼ��ˮ��֮��Ϻ�����������к͡�ϡ�;�����������ˮ�����ɵ��ᣨH+����
�ʴ�Ϊ��2H2SO3+O2=2H2SO4���к͡�ϡ�;�����������ˮ�����ɵ��ᣨH+����
��2���ɶ��������뽬Һ�е�̼����Լ�����Ŀ�����Ӧ����ʯ���֪��
�÷�ӦΪ2CaCO3+2SO2+O2+4H2O�T2CaSO4?2H2O+2CO2��
�ʴ�Ϊ��2CaCO3+2SO2+O2+4H2O�T2CaSO4?2H2O+2CO2��
��3���ɱ����е����ݿ�֪���ܶȻ�ԽС��Խ��ת��Ϊ��������Ȼֻ��������ܶȻ�С����Ӧѡ�����ƣ�
�ʴ�Ϊ��B��
��4����Pb��OH��2���ܶȻ�Ϊ1.2��10-15��pH=8.0��c��OH-��=10-6mol?L-1����c��Pb2+��=
=1.2��10-3mol?L-1������Ҫ����ˮ�ۺ��ŷű�Ϊc��Pb2+������1.0��l0-8mol?L-1��
��Ȼ1.2��10-3mol?L-1��1.0��l0-8mol?L-1�������ŷű����ʴ�Ϊ��1.2��10-3mol?L-1����
���ɿ����е�������H2SO3����Ϊ���ᣬ�÷�ӦΪ2H2SO3+O2=2H2SO4��������ġ���ˮ����Ҫ�ô�������Ȼ��ˮ��֮��Ϻ�����������к͡�ϡ�;�����������ˮ�����ɵ��ᣨH+����
�ʴ�Ϊ��2H2SO3+O2=2H2SO4���к͡�ϡ�;�����������ˮ�����ɵ��ᣨH+����
��2���ɶ��������뽬Һ�е�̼����Լ�����Ŀ�����Ӧ����ʯ���֪��
�÷�ӦΪ2CaCO3+2SO2+O2+4H2O�T2CaSO4?2H2O+2CO2��
�ʴ�Ϊ��2CaCO3+2SO2+O2+4H2O�T2CaSO4?2H2O+2CO2��
��3���ɱ����е����ݿ�֪���ܶȻ�ԽС��Խ��ת��Ϊ��������Ȼֻ��������ܶȻ�С����Ӧѡ�����ƣ�
�ʴ�Ϊ��B��
��4����Pb��OH��2���ܶȻ�Ϊ1.2��10-15��pH=8.0��c��OH-��=10-6mol?L-1����c��Pb2+��=
| Ksp |
| c(OH-)2 |
��Ȼ1.2��10-3mol?L-1��1.0��l0-8mol?L-1�������ŷű����ʴ�Ϊ��1.2��10-3mol?L-1����
���������⿼����Ⱦ�Ĵ�����������ij���ת����������ѧ֪ʶ���ϰ���е���Ϣ���ɽ��ϰ����ͼ�������ǽ����Ĺؼ����ϺõĿ���ѧ���������⡢��������������

��ϰ��ϵ�д�
�����Ŀ
�����ű�����ȫ����̡�ʡ��֮�Ƶĸ�����������ȴƵƵ�ܵ���������ţ�������ˮԴ��ˮ��״�����棬����9�������е�34������ʽ��������ˮԴ��ˮ�ʴ����Ϊ73.3%��������ͬ�������½������Ա������������ǿ̲��ݻ������Σ�
�����������ǿ��ƶ���������Ⱦ����Ҫ�����ֶΣ�
��1�����ú�ˮ������һ����Ч�ķ������乤��������ͼ1��ʾ��
ij�о�С��Ϊ̽����ߺ���������SO2������Ч�ʵĴ�ʩ����������Ȼ��ˮ��pH��8�����պ���������ģ��ʵ�飬ʵ������ͼ2��ʾ��
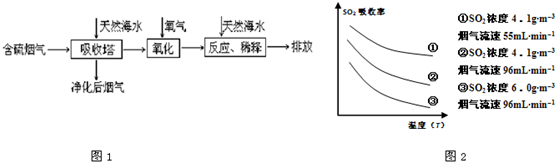
�ٸ���ͼʾʵ������Ϊ�����һ��Ũ�Ⱥ���������SO2������Ч�ʣ����д�ʩ��ȷ����______��������ĸ��ţ�
A������ͨ�뺬���������¶ȡ���������B����Сͨ�뺬������������
C��������Ȼ��ˮ�Ľ����������������� D������Ȼ��ˮ�м�����ʯ��
����Ȼ��ˮ�����˺��������������H2SO3��ʹ�ÿ����е���������������д���÷�Ӧ�Ļ�ѧ����ʽ��______��������ġ���ˮ����Ҫ�ô�������Ȼ��ˮ��֮��Ϻ�����ŷţ��ò�������ҪĿ����______��
��2��ʯ��ʯ-ʯ��ʪ�����������ռ����Ĺ���ԭ���������еĶ��������뽬Һ�е�̼����Լ�����Ŀ�����Ӧ����ʯ�ࣨCaSO4?2H2O����д���÷�Ӧ�Ļ�ѧ����ʽ��______��
���ؽ������ӶԺ������������������Ⱦ��ij��������ˮ��pH��2���к���Ag+��Pb2+���ؽ������ӣ���Ũ�ȸ�ԼΪ0.01mol?L-1���ŷ�ǰ���ó�������ȥ���������ӣ������й��������£�
| ���ܵ���� | AgI | AgOH | Ag2S | PbI2 | Pb��OH��2 | PbS |
| Ksp | 8.3��10-17 | 5.6��10-8 | 6.3��10-50 | 7.1��10-9 | 1.2��10-15 | 3.4��10-28 |
A��NaOH����B��Na2S����C��KI�� D��Ca��OH��2
��4�����ֻ����ʯ�Ҵ�������Pb2+�ķ�ˮ��ʹ��Һ��pH=8.0��������ķ�ˮ�У�c��Pb2+��=______��������Ҫ����ˮ�ۺ��ŷű�Ϊc��Pb2+������1.0��l0-8mol?L-1���ʴ�����ķ�ˮ�Ƿ�����ŷű���______ ����ǡ�����
�����ű�����ȫ����̡�ʡ��֮�Ƶĸ�����������ȴƵƵ�ܵ���������ţ�������ˮԴ��ˮ��״�����棬����9�������е�34������ʽ��������ˮԴ��ˮ�ʴ����Ϊ73.3%��������ͬ�������½������Ա������������ǿ̲��ݻ������Σ�
�����������ǿ��ƶ���������Ⱦ����Ҫ�����ֶΣ�
��1�����ú�ˮ������һ����Ч�ķ������乤��������ͼ1��ʾ��
ij�о�С��Ϊ̽����ߺ���������SO2������Ч�ʵĴ�ʩ����������Ȼ��ˮ��pH��8�����պ���������ģ��ʵ�飬ʵ������ͼ2��ʾ��
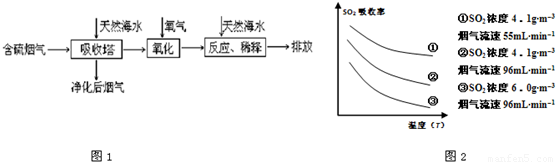
�ٸ���ͼʾʵ������Ϊ�����һ��Ũ�Ⱥ���������SO2������Ч�ʣ����д�ʩ��ȷ����______��������ĸ��ţ�
A������ͨ�뺬���������¶� B����Сͨ�뺬������������
C��������Ȼ��ˮ�Ľ����� D������Ȼ��ˮ�м�����ʯ��
����Ȼ��ˮ�����˺��������������H2SO3��ʹ�ÿ����е���������������д���÷�Ӧ�Ļ�ѧ����ʽ��______��������ġ���ˮ����Ҫ�ô�������Ȼ��ˮ��֮��Ϻ�����ŷţ��ò�������ҪĿ����______��
��2��ʯ��ʯ-ʯ��ʪ�����������ռ����Ĺ���ԭ���������еĶ��������뽬Һ�е�̼����Լ�����Ŀ�����Ӧ����ʯ�࣮д���÷�Ӧ�Ļ�ѧ����ʽ��______��
���ؽ������ӶԺ������������������Ⱦ��ij��������ˮ��pH��2���к���Ag+��Pb2+���ؽ������ӣ���Ũ�ȸ�ԼΪ0.01mol?L-1���ŷ�ǰ���ó�������ȥ���������ӣ������й��������£�
��3������Ϊ����ˮ��Ͷ��______������ĸ��ţ�������Ч����ã�
A��NaOH B��Na2S C��KI D��Ca��OH��2
��4�����ֻ����ʯ�Ҵ�������Pb2+�ķ�ˮ��ʹ��Һ��pH=8.0��������ķ�ˮ�У�c��Pb2+��=______��������Ҫ����ˮ�ۺ��ŷű�Ϊc��Pb2+������1.0×l0-8mol?L-1���ʴ�����ķ�ˮ�Ƿ�����ŷű���______ ����ǡ�����
�����������ǿ��ƶ���������Ⱦ����Ҫ�����ֶΣ�
��1�����ú�ˮ������һ����Ч�ķ������乤��������ͼ1��ʾ��
ij�о�С��Ϊ̽����ߺ���������SO2������Ч�ʵĴ�ʩ����������Ȼ��ˮ��pH��8�����պ���������ģ��ʵ�飬ʵ������ͼ2��ʾ��

�ٸ���ͼʾʵ������Ϊ�����һ��Ũ�Ⱥ���������SO2������Ч�ʣ����д�ʩ��ȷ����______��������ĸ��ţ�
A������ͨ�뺬���������¶� B����Сͨ�뺬������������
C��������Ȼ��ˮ�Ľ����� D������Ȼ��ˮ�м�����ʯ��
����Ȼ��ˮ�����˺��������������H2SO3��ʹ�ÿ����е���������������д���÷�Ӧ�Ļ�ѧ����ʽ��______��������ġ���ˮ����Ҫ�ô�������Ȼ��ˮ��֮��Ϻ�����ŷţ��ò�������ҪĿ����______��
��2��ʯ��ʯ-ʯ��ʪ�����������ռ����Ĺ���ԭ���������еĶ��������뽬Һ�е�̼����Լ�����Ŀ�����Ӧ����ʯ�࣮д���÷�Ӧ�Ļ�ѧ����ʽ��______��
���ؽ������ӶԺ������������������Ⱦ��ij��������ˮ��pH��2���к���Ag+��Pb2+���ؽ������ӣ���Ũ�ȸ�ԼΪ0.01mol?L-1���ŷ�ǰ���ó�������ȥ���������ӣ������й��������£�
| ���ܵ���� | AgI | AgOH | Ag2S | PbI2 | Pb��OH��2 | PbS |
| Ksp | 8.3×10-17 | 5.6×10-8 | 6.3×10-50 | 7.1×10-9 | 1.2×10-15 | 3.4×10-28 |
A��NaOH B��Na2S C��KI D��Ca��OH��2
��4�����ֻ����ʯ�Ҵ�������Pb2+�ķ�ˮ��ʹ��Һ��pH=8.0��������ķ�ˮ�У�c��Pb2+��=______��������Ҫ����ˮ�ۺ��ŷű�Ϊc��Pb2+������1.0×l0-8mol?L-1���ʴ�����ķ�ˮ�Ƿ�����ŷű���______ ����ǡ�����