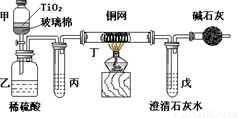
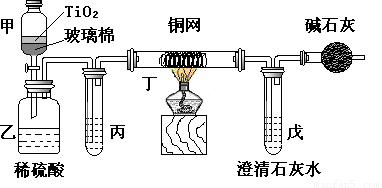
��Ŀ����
. ��Ƴ������������ƹ��գ����������Ƶķ�ˮʱ�����ڴ���TiO2�����£�����NaClO��CN������������CNO�����������������¼�����NaClO������N2��CO2������������Ա���ܱ�ϵͳ������ͼװ�ý���ʵ�飬��֤��������������Ч�ԣ���ͨ���ⶨ������̼����ȷ��CN���������İٷ��ʡ�

��Ũ����CN�����ӵ���ˮ�����NaClO��Һ�Ļ��Һ��200 mL������CN����Ũ��Ϊ0.05 mol��L��1��
������У�������Ƥ����һ��ʱ�����Ƥ���ͻ�����ʹ��Һȫ���������У��رջ������ش��������⣺
�� ���з�Ӧ�����ӷ���ʽΪ ��
���з�Ӧ�����ӷ���ʽΪ ��
�� �������ɵ������N2��CO2�⣬����HCl��������Cl2�ȡ����м���ij����Լ��DZ���ʳ��ˮ���������� ��
����ʵ���е������� ��
װ�м�ʯ�ҵĸ���ܵ������� ��
�� ����ʢ�к�Ca(OH)2 0.02mol��ʯ��ˮ����ʵ�������й�����0.82 g���������ʵ���в��CN���������İٷ��ʵ��� ���ò��ֵ�빤ҵʵ�ʴ����İٷ����������ƫ�ͣ���Ҫ˵�����ܵ�ԭ�� ��
�� ��
�����һ�������ȷ�ȵĽ��飨Ҫ�пɲ����ԣ��Ҳ�����
��
(��15��)
�� CN��+ClO��CNO��+Cl����
2CNO��+2H+ +3ClO��N2��+2CO2��+3Cl��+H2O
�� ��ȥHCl���� ȥ��Cl2 ��ֹ������CO2��������Ӱ��ⶨȷ��
�� 82%
��װ���ҡ��������п���������CO2��
��CO2�������ٶȽϿ�δ�����еij���ʯ��ˮ��ַ�Ӧ ��
��Cl2��HCl�ڱ�������δ������ȫ��
(��������������㼴�ɣ�ÿ���һ���1�֣���2��)
����һ������������Һһ��ȫ���������У���Ϊ�ִμ��룬����CO2�IJ����ٶȣ�
�����������ƿ����Ϊ�����������ӵ��Ǹ����в���һ�����ܵ�Һ�����£���Ӧ������ͨ���ȥCO2�Ŀ�����ʹװ����������CO2�����ܶ����Ca(OH)2��Ӧ��
�������������г���ʯ��ˮ��ΪŨ�Ƚϴ��NaOH��Һ����Ӧ�����������м�������CaCl2�������������ȵ�
(�����������һ�㼴��) ��ÿ��2�֣���15�֣�

 ��ĩ���ƾ�ϵ�д�
��ĩ���ƾ�ϵ�д� ���ɿ��ñ���ϵ�д�
���ɿ��ñ���ϵ�д�