��Ŀ����
��֪��������ͨ����������S8(б����)����ʽ���ڣ���������״̬ʱ������S2��S4��S6��S8�ȶ���ͬ�������壬����S4��S6��S8�������ƵĽṹ�ص㣬��ṹ����ͼ��ʾ��
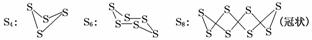
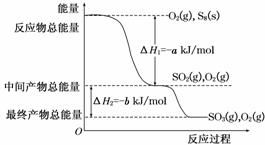
��һ�������£�S8(s)��O2(g)������Ӧ����ת��ΪSO2(g)��SO3(g)����Ӧ���̺�������ϵ������ͼ��ʾ(ͼ�еĦ�H��ʾ����1 mol���������)��
(1)д����ʾS8ȼ���ȵ��Ȼ�ѧ����ʽ_________________________________
____________��
(2)д��SO3�ֽ�����SO2��O2���Ȼ�ѧ����ʽ______________________________
________________________________________________________________________��
(3)��ѧ�Ϲ涨�����γ�1 mol��ѧ�����ջ�ų���������Ϊ�û�ѧ���ļ��ܣ���λkJ��mol������֪�������ļ���Ϊd kJ��mol��1���������ļ���Ϊe kJ��mol��1����S8������������ļ���Ϊ____________________________________��
������(1)ȼ����ָ����1 molȼ����ȫȼ�������ȶ�������ʱ�ų�����������˸��ݶ�����д����ѧ����ʽ��S8(s)��8O2(g) ,��8SO2(g)������������и������ʱ���ָ����1 mol��������ݣ���˸÷�Ӧ���Ȼ�ѧ����ʽΪS8(s)��8O2(g)===8SO2(g)����H����8a kJ��mol��1��
(2)�ɸ�������ͼʾֱ��д���Ȼ�ѧ����ʽ��
(3)��S8������������ļ���Ϊx kJ��mol��1�����ڷ�Ӧ�ȵ��ڷ�Ӧ��ļ���֮�ͼ�ȥ������ļ���֮�ͣ���Ϣٿɵ�8x��8e��16d����8a�����x��2d��a��e��
�𰸣�(1)S8(s)��8O2(g)===8SO2(g)����H����8a kJ��mol��1
(2)2SO3(g)===2SO2(g)��O2(g)����H����2b kJ��mol��1
(3)(2d��a��e) kJ��mol��1

���п���������SO2��CO2������ǣ� ��
�ٳ����ʯ��ˮ�����������ˮ�����Ը�����آ��Ȼ�����Ʒ����Һ
| A���٢ܢݢ� | B���ڢۢܢ� | C���٢ڢۢ� | D���ڢۢܢ� |

 ��Cl����CO
��Cl����CO ��SO
��SO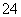 ���ֱַ�ȡ100 mL�����ȷ���Һ��������ʵ�飺
���ֱַ�ȡ100 mL�����ȷ���Һ��������ʵ�飺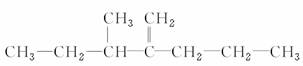 ����������Ϊ_______________________________________________________________��
����������Ϊ_______________________________________________________________��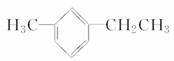 ��������Ϊ______________
��������Ϊ______________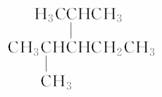 ��������Ϊ_______________
��������Ϊ_______________