��Ŀ����
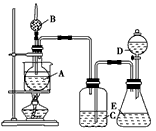
ijѧ���������ͼ��ʾ��װ�ý���ʵ�飬��װ������װ�Լ���ʵ�������ʵ���������£�
��.װ������װ���Լ��ǣ���Aƿװ��ˮ�Ҵ����ڷ���ˮ��X����B�������װ��ʯ�ң���C��D�ж�װŨ�����Eƿ��װ���Լ�Y
��.ʵ������������ǣ���ˮԡ����Aƿ����D��Ũ���Ỻ������E�����Լ�Y���ã�����C�е����д������ݷų���Aƿ��X��ɫ����B�ܻӷ���������ɵ�ȼ��
��.װ������װ���Լ��ǣ���Aƿװ��ˮ�Ҵ����ڷ���ˮ��X����B�������װ��ʯ�ң���C��D�ж�װŨ�����Eƿ��װ���Լ�Y
��.ʵ������������ǣ���ˮԡ����Aƿ����D��Ũ���Ỻ������E�����Լ�Y���ã�����C�е����д������ݷų���Aƿ��X��ɫ����B�ܻӷ���������ɵ�ȼ��
��ش��������⣺
(1)Eƿ����װ���Լ�Y��________(��д���)
a������ʳ��ˮ��b��MnO2��NaCl�Ļ���c��Ũ����
(2)C��ŨH2SO4�����������_______________________��Dƿ��ŨH2SO4�����������________________________________��
(3)Aƿ�з�����Ӧ�Ļ�ѧ����ʽ��______________________����Ӧ������__________�������ɵ�________________(д����)��B���ڴ���ȼ��
(4)��ˮ��X��ѡ��____________��������ָʾ�����õ�ԭ����______________________��
(5)��ʵ����֤���Ҵ������к�����ԭ�ӵ�������________________________��
(6)�����װ���е�Cƿȥ����ʵ��Ŀ���Ƿ��ܹ��ﵽ��_____(��ܡ����ܡ�)����Ϊ___________________________��
(1)Eƿ����װ���Լ�Y��________(��д���)
a������ʳ��ˮ��b��MnO2��NaCl�Ļ���c��Ũ����
(2)C��ŨH2SO4�����������_______________________��Dƿ��ŨH2SO4�����������________________________________��
(3)Aƿ�з�����Ӧ�Ļ�ѧ����ʽ��______________________����Ӧ������__________�������ɵ�________________(д����)��B���ڴ���ȼ��
(4)��ˮ��X��ѡ��____________��������ָʾ�����õ�ԭ����______________________��
(5)��ʵ����֤���Ҵ������к�����ԭ�ӵ�������________________________��
(6)�����װ���е�Cƿȥ����ʵ��Ŀ���Ƿ��ܹ��ﵽ��_____(��ܡ����ܡ�)����Ϊ___________________________��
(1)c��
(2)����HCl���壻����Ũ�����л��е�ˮ�֣�ŨH2SO4����ˮ���ȣ�������HCl�����ݳ���
(3)CH3CH2OH��HCl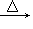 CH3CH2Cl��H2O��ȡ����Ӧ�������顡
CH3CH2Cl��H2O��ȡ����Ӧ�������顡
(4)��ˮCuSO4��ʵ������й۲쵽��ˮCuSO4�ɰױ�����˵����Ӧ����ˮ���ɣ���CuSO4�������
CuSO4��5H2O��
(5)��ˮCuSO4����֤���˷�Ӧ��һ����ˮ���ɣ�ˮ�е���Ԫ�ز���������HCl����ֻ�����Ҵ��ṩ��
(6)���ܣ�HCl�ӷ�ʱ����ˮ������������ȥ�����ж�ʹ��ˮCuSO4������ˮ�Ƿ��������Ҵ�
(2)����HCl���壻����Ũ�����л��е�ˮ�֣�ŨH2SO4����ˮ���ȣ�������HCl�����ݳ���
(3)CH3CH2OH��HCl
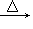 CH3CH2Cl��H2O��ȡ����Ӧ�������顡
CH3CH2Cl��H2O��ȡ����Ӧ�������顡(4)��ˮCuSO4��ʵ������й۲쵽��ˮCuSO4�ɰױ�����˵����Ӧ����ˮ���ɣ���CuSO4�������
CuSO4��5H2O��
(5)��ˮCuSO4����֤���˷�Ӧ��һ����ˮ���ɣ�ˮ�е���Ԫ�ز���������HCl����ֻ�����Ҵ��ṩ��
(6)���ܣ�HCl�ӷ�ʱ����ˮ������������ȥ�����ж�ʹ��ˮCuSO4������ˮ�Ƿ��������Ҵ�

��ϰ��ϵ�д�
 ��ʦָ����ĩ��̾�ϵ�д�
��ʦָ����ĩ��̾�ϵ�д�
�����Ŀ
 ijѧ���������ͼ��ʾ��װ�ý���ʵ�飬��װ������װ�Լ���ʵ�������ʵ���������£�
ijѧ���������ͼ��ʾ��װ�ý���ʵ�飬��װ������װ�Լ���ʵ�������ʵ���������£�
 �����Ǽ�����Ҫ�Ļ���ԭ�ϣ���ҵ�Ͻ�������NO2���ܱ���������ˮ��η���ѭ�������Ʊ����ᣮ
�����Ǽ�����Ҫ�Ļ���ԭ�ϣ���ҵ�Ͻ�������NO2���ܱ���������ˮ��η���ѭ�������Ʊ����ᣮ
