��Ŀ����
���ž��÷�չ����������ˮƽ����ߣ���������ʡ����ӵ�����ʽϿ��������ơ�
�� �ұ���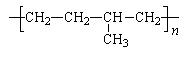 ���������Ժö����������������Ŵ��ܷ⽺������ҵ�����ұ��ĵ����������������������� ��
���������Ժö����������������Ŵ��ܷ⽺������ҵ�����ұ��ĵ����������������������� ��
(2) �ڷ���������Ԫ��ת�������β�����а�װһ����Ϊ��̽�����ĵ绯ѧԪ�����ɲⶨ�ˣ���=��ڴ��Ŀ�����/��ȫȼ����Ҫ�Ŀ�������ֵ������ͼ��ʾ��
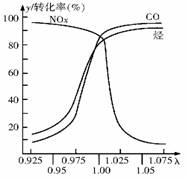
�������ٶ�����ʱ����ڴ��Ŀ���������ʱ�ų�β���е���Ҫ��Ⱦ������������ ����ת������ǰ�벿��һ����̼�͵������NOx��ͨ������������Ӧת��Ϊ����Ⱦ�����壬�䷴Ӧ�Ļ�ѧ����ʽΪ������������������������ ��
��3���ⶨ����β����NO��NO2�ķ���֮һ����3����H2O2��Һ��������HNO3������NaOH����Һ�ζ�����Ҫȷ��β����NO��NO2��������ܺͣ������������___________(ѡ�����)��
A��������Ʒ�����������������������
B��NaOH����Һ��Ũ�ȼ������ĵ����
C���ӵζ���ʼ��ָʾ����ɫ�����ʱ�䡡����
D�����ֵ�������������
(4)Ϊ�˽�������β���Դ�������Ⱦ���йز������ü״������Ϊ��������ȼ�ϡ�д���úϳ���(CO��H2)�����״��Ļ�ѧ����ʽ__________________________���ø÷����ϳɼ״�������ŵ���______________________________________________����֪�״�ȼ����Ϊ726kJ/mol����д���״�ȼ�յ��Ȼ�ѧ����ʽ������������ ��������
��CH3�DCH=CH2, ��1�֣�CH2=CH2 ��1�֣�
(2)NOX ��1�֣� 2xCO+2NOX=N2+2xCO2 ��1�֣�
(3)AB��2�֣�
(4) ��CO��2H2��CH3OH��1�֣� ԭ��������Ϊ100����1�֣�
2CH3OH(l)+3O2(g) =2CO2(g)+4H2O(l) ��H=-1452kJ/mol��2�֣�

 ��Ч���ܿ�ʱ��ҵϵ�д�
��Ч���ܿ�ʱ��ҵϵ�д� �ݾ�ѵ������ϵ�д�
�ݾ�ѵ������ϵ�д� С����ȫ�ܼ��ϵ�д�
С����ȫ�ܼ��ϵ�д� ��2010?�ij�һģ�����ž��÷�չ��������Խ��Խ�࣬ͬʱҲ�����˽�ͨӵ���Ϳ�����Ⱦ������β��װ��������ڴ�������������������õĹ�����ͼ��ʾ������˵����ȷ���ǣ�������
��2010?�ij�һģ�����ž��÷�չ��������Խ��Խ�࣬ͬʱҲ�����˽�ͨӵ���Ϳ�����Ⱦ������β��װ��������ڴ�������������������õĹ�����ͼ��ʾ������˵����ȷ���ǣ�������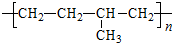 ���������Ժö����������������Ŵ��ܷ⽺������ҵ�����ұ��ĵ�����
���������Ժö����������������Ŵ��ܷ⽺������ҵ�����ұ��ĵ�����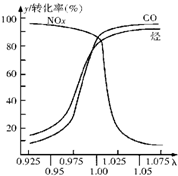


 CO+N2
CO+N2