��Ŀ����
����Ŀ����50mLNaOH��Һ����������ͨ��һ������CO2�����ȡ����Һ10mL����ϡ�͵�100mL�������ϡ�ͺ����Һ����μ���0.1mol/L��HCl��Һ��������CO2�����������״���£����������HCl�����֮��Ĺ�ϵ��ͼ��ʾ���Է�����
��1��NaOH������CO2������ڼס��� ��������£�������Һ�д��ڵ�������
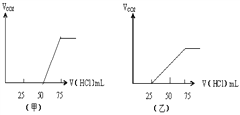
�ף�_____________�����ʵ���֮���ǣ�__________
�ң�_____________�����ʵ���֮���ǣ�__________
��2������������²�����CO2���壨��״�������Ǽף�___________���ң�___________��
��3��ԭNaOH��Һ�����ʵ���Ũ����___________��
���𰸡� NaOH��Na2CO3 1:1 NaHCO3��Na2CO3 1:1 56mL 112mL 0.75mol/L
�����������������̼���������ᷴӦʱ���������У��ֱ���CO32-+H+=HCO3-��HCO3-+H+=H2O+CO2�������������������������ͬ���ɼ�ͼ��֪��ϡ�ͺ����Һ����μ����һ��25mL����ʱ���������Ʒ�Ӧ���ڶ���25mL����ʱ����CO32-+H+=HCO3-��������25mL����ʱ����HCO3-+H+=H2O+CO2����������Һ�е�����ΪNaOH��Na2CO3�������ʵ���֮����1:1����ͼ�ҿ�֪��ϡ�ͺ����Һ����μ����һ��25mL����ʱ����CO32-+H+=HCO3-������ڶ��ݺ͵�����25mL����ʱ����HCO3-+H+=H2O+CO2����������Һ�е�������NaHCO3��Na2CO3�������ʵ���֮����1:1��
��1��NaOH������CO2������ڼס��� ��������£�������Һ�д��ڵ�������
�ף�NaOH��Na2CO3�������ʵ���֮���ǣ�1:1��
�ң�NaHCO3��Na2CO3�����ʵ���֮���ǣ�1:1��
��2������������²�����CO2���壨��״����������������HCO3-+H+=H2O+CO2����n(CO2)=n(HCl)=0.025L![]() 0.1mol/L=0.0025mol�����Ϊ0.0025mol
0.1mol/L=0.0025mol�����Ϊ0.0025mol ![]() 22.4L/mol=0.056L=56mL��������������Ĺ�������HCO3-+H+=H2O+CO2�������������Ǽ�2������������CO2����112mL��
22.4L/mol=0.056L=56mL��������������Ĺ�������HCO3-+H+=H2O+CO2�������������Ǽ�2������������CO2����112mL��
��3������ͼ���֪��50mLNaOH��Һ���ն�������75mL0.1mol/L�����������Ȼ�����Һ���ڷ�Ӧ�������������غ㡢�������غ㣬����ԭNaOH��Һ�����ʵ���Ũ����![]() 0.75mol/L��
0.75mol/L��

����Ŀ���⼰�仯�����ںϳ�ɱ������ҩ��ȷ�����й㷺��;���ش��������⣺
��1�������ĵ⸻���ں����У���ˮ��ȡ��Ũ��������Ũ��Һ�м�MnO2��H2SO4�����ɵõ�I2���÷�Ӧ�Ļ�ԭ����Ϊ______��
��2����֪��Ӧ2HI(g)![]() H2(g)+I2(g)����H = +11 kJ��mol��1��l molH2(g)��1 mol I2(g)�����л�ѧ������ʱ�ֱ���Ҫ����436 kJ��151 kJ����������1 mol HI(s)�����л�ѧ������ʱ�����յ�����Ϊ______kJ��
H2(g)+I2(g)����H = +11 kJ��mol��1��l molH2(g)��1 mol I2(g)�����л�ѧ������ʱ�ֱ���Ҫ����436 kJ��151 kJ����������1 mol HI(s)�����л�ѧ������ʱ�����յ�����Ϊ______kJ��
��3��Bodensteins�о������з�Ӧ��2HI(g)![]() H2(g)+I2(g)����716 Kʱ�����������е⻯������ʵ�������x(HI)�뷴Ӧʱ��t�Ĺ�ϵ���±���
H2(g)+I2(g)����716 Kʱ�����������е⻯������ʵ�������x(HI)�뷴Ӧʱ��t�Ĺ�ϵ���±���
t/min | 0 | 20 | 40 | 60 | 80 | 120 |
x(HI) | 1 | 0.91 | 0.85 | 0.815 | 0.795 | 0.784 |
x(HI) | 0 | 0.60 | 0.73 | 0.773 | 0.780 | 0.784 |
�ٸ�������ʵ�������÷�Ӧ��ƽ�ⳣ��K�ļ���ʽΪ__________________��
��������Ӧ�У�����Ӧ����Ϊ![]() ��=k��x2(HI)���淴Ӧ����Ϊ
��=k��x2(HI)���淴Ӧ����Ϊ![]() ��=k��x(H2)x(I2)������k����k��Ϊ���ʳ�������k��Ϊ____________________(��K��k����ʾ)����k��=0.0027 min��1����t = 40 minʱ��
��=k��x(H2)x(I2)������k����k��Ϊ���ʳ�������k��Ϊ____________________(��K��k����ʾ)����k��=0.0027 min��1����t = 40 minʱ�� ![]() �� = _______________min��1��
�� = _______________min��1��
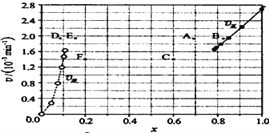
��������ʵ�����ݼ���õ�![]() ����x(HI)��
����x(HI)��![]() ����x(H2)�Ĺ�ϵ������ͼ��ʾ�������ߵ�ijһ�¶�ʱ����Ӧ���´ﵽƽ�⣬��Ӧ�ĵ�ֱ�Ϊ______________(����ĸ)��
����x(H2)�Ĺ�ϵ������ͼ��ʾ�������ߵ�ijһ�¶�ʱ����Ӧ���´ﵽƽ�⣬��Ӧ�ĵ�ֱ�Ϊ______________(����ĸ)��