��Ŀ����
����Ŀ��������ʡ����ʦ����ѧ��һ������ѧ2017�����5��Ѻ�⿼�����ۻ�ѧ���⡿2011�꣬��ʡ����ѧԺ�����Ŷ��з���һ��������Ҷ������һƬ�뵼����ϼ�����������Ĵ������м䡣����������Ҷ�����������Һ�У���������������Ҷ�ϣ��ͻ�ֽ�ˮ����H2��O2���ṹ��ͼ��ʾ�������й�������Ҷ˵����ȷ����
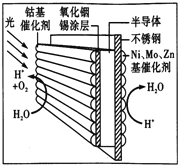
A. ��װ�����ս�̫����ת��Ϊ����
B. Ni��Mo��Zn��������Ϊ����������������Ӧ
C. ���Խ�������Ҷ�����ڼ�����Һ��
D. �ܻ������Ϸ�����Ӧ��2H2O-4e-=4H++O2��
���𰸡�D
��������A. ��װ�����ս�̫����ת��Ϊ���ܣ���ת��Ϊ��ѧ�ܣ���A����B. Ni��Mo��Zn��������Ϊ��������������Ӧ��H�� �õ��ӣ�������ԭ��Ӧ����B����C. �����Խ�������Ҷ�����ڼ�����Һ�У����������£��������ܲ���H�� ����C����D. �ܻ������ڸ�������������������Ӧ��������Ӧ��2H2O-4e-=4H++O2������D��ȷ����ѡD��

 ���Ͱ�ͨ��ĩ���ϵ�д�
���Ͱ�ͨ��ĩ���ϵ�д�����Ŀ�����Ȼ���(PCl5)���л��ϳ�����Ҫ���Ȼ���,���������Ȼ���(PCl3)�Ȼ��õ�:
PCl3(g)+Cl2(g)![]() PCl5(g)����H=-93.0 kJ��mol-1��ij�¶���,���ݻ��㶨Ϊ2.0 L���ܱ������г���2.0 mol PCl3��1.0 mol Cl2,һ��ʱ���Ӧ��ƽ��״̬,ʵ���������±���ʾ:
PCl5(g)����H=-93.0 kJ��mol-1��ij�¶���,���ݻ��㶨Ϊ2.0 L���ܱ������г���2.0 mol PCl3��1.0 mol Cl2,һ��ʱ���Ӧ��ƽ��״̬,ʵ���������±���ʾ:
t/s | 0 | 50 | 150 | 250 | 350 |
n(PCl5)/mol | 0 | 0.24 | 0.36 | 0.40 | 0.40 |
(1)0��150 s�ڵ�ƽ����Ӧ����v(PCl3)=________mol��L-1��s-1��
(2)���¶���,�˷�Ӧ�Ļ�ѧƽ�ⳣ������ֵΪ____(���÷�����ʾ)��
(3)��Ӧ��250 sʱ,�÷�Ӧ�ų�������Ϊ________kJ��
(4)���й���������Ӧ��˵����,��ȷ����______(����ĸ)��
a.�����Ƿ��ƽ��״̬,���������ܶ�ʼ�ղ���
b.300 sʱ,�����¶�,�����淴Ӧ����ͬʱ����
c.350 sʱ,����ܱ������г��뵪��,ƽ��������Ӧ�����ƶ�
d.��ͬ������,����ʼ����ܱ������г���1.0 mol PCl3��0.2 mol Cl2,�ﵽ��ѧƽ��״̬ʱ,n(PCl5)<0.20 mol
(5)�¶�T1ʱ,���������PCl5�����������ʱ��t�仯��ʾ��ͼ���ϡ�����������ͬ,������ͼ�л����¶�ΪT2(T2>T1)ʱ,PCl5�����������ʱ��仯������________��

