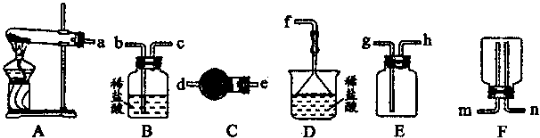
��Ŀ����
��18.4mol/L��Ũ��������100mLŨ��Ϊ1mol/L��ϡ���ᣬ������ɷ�Ϊ���¸������뽫�����пհ״���д��������
A������Ͳ��ȡ
B����Լ30mL����ˮ������ϴ���ձ��Ͳ���������ÿ�ε�ϴҺ����������ƿ��
C����ϡ�ͺ������С�ĵ�������ƿ��
D�����100mL����ƿ���Ƿ�©ˮ
E���ò���������������ˮ��������ƿ����Һ��ӽ�����
F���ǽ�ƿ���������ߵ���ҡ����Һ
G���ý�ͷ�ι�������ƿ����μ�������ˮ����Һ����͵�ǡ�����������
��ݴ���д��
��1����ȷ�IJ���˳���ǣ�����ĸ��д����
��2������A����������
��3����ʵ������ϡ���ᣬ������������ͷ�ιܣ��Լ�ƿ�⣬�������Ҫ�����У�
��4�����в�����ʹ������ҺŨ��ƫ�͵���
A��ת����Һ��û��ϴ���ձ��ڱ�
B������ǰ����ƿ��������ˮ
C������ʱ����
D��������Һδ����ȴ��ת�Ƶ�����ƿ��ȥ��
A������Ͳ��ȡ
5.4
5.4
mLŨ���ᣬ����ע��װ��Լ50mL����ˮ���ձ�����ò���������B����Լ30mL����ˮ������ϴ���ձ��Ͳ���������ÿ�ε�ϴҺ����������ƿ��
C����ϡ�ͺ������С�ĵ�������ƿ��
D�����100mL����ƿ���Ƿ�©ˮ
E���ò���������������ˮ��������ƿ����Һ��ӽ�����
1-2cm
1-2cm
��F���ǽ�ƿ���������ߵ���ҡ����Һ
G���ý�ͷ�ι�������ƿ����μ�������ˮ����Һ����͵�ǡ�����������
��ݴ���д��
��1����ȷ�IJ���˳���ǣ�����ĸ��д����
DACBEGF
DACBEGF
����2������A����������
��ȴ������
��ȴ������
����ܽ���C��������3����ʵ������ϡ���ᣬ������������ͷ�ιܣ��Լ�ƿ�⣬�������Ҫ�����У�
��Ͳ���ձ���100mL����ƿ
��Ͳ���ձ���100mL����ƿ
����4�����в�����ʹ������ҺŨ��ƫ�͵���
AC
AC
����ѡ����ţ�A��ת����Һ��û��ϴ���ձ��ڱ�
B������ǰ����ƿ��������ˮ
C������ʱ����
D��������Һδ����ȴ��ת�Ƶ�����ƿ��ȥ��
����������ϡ��ǰ�����ʵ����ʵ����������Ũ��������������ʵ������ľ��岽����н������е���գ�
��1������ʵ������IJ�����в���˳������
��2������ʵ������IJ�����н��
��3������ʵ������IJ���ȷ���������������
��4����������������n��V��Ӱ�죬���c=
�����������������Ũ�ȵ�Ӱ�죮
��1������ʵ������IJ�����в���˳������
��2������ʵ������IJ�����н��
��3������ʵ������IJ���ȷ���������������
��4����������������n��V��Ӱ�죬���c=
| n |
| V |
����⣺��Ũ��������ΪVml����18.4mol/L��Vml=1mol/L��100ml�����V=5.4����ˮ��Һ�����̶���1��2cmʱ�����ý�ͷ�ιܵμӣ�ʹ��Һ�İ�Һ�����͵��������ƽ��
�ʴ�Ϊ5.4��1��2cm��
��1�����Ʋ�������ȡ��ϡ�͡���Һ��ϴ�ӡ����ݡ�ҡ�ȵȲ�����һ������Ͳ��ȡ���õ���ͷ�ιܣ����������ձ���ϡ�ͣ���ȴ��ת�Ƶ�100mL����ƿ�У����ò�����������ת����ϣ�����������ˮϴ���ձ���������2��3�β���ϴ��Һȫ��ת�Ƶ�����ƿ�У��ټ���������ˮ������ˮ��Һ�����̶���1��2cmʱ�����ý�ͷ�ιܵμӣ�ʹ��Һ�İ�Һ�����͵��������ƽ������ƿ�����������µߵ�ҡ�ȣ�
�ʴ�Ϊ��DACBEGF��
��2���ɣ�1�����������֪��ϡ�ͺ������ȴ�����£���ת�Ƶ�����ƿ�У�
�ʴ�Ϊ����ȴ�����£�
��3���ɣ�1�����������֪�������������ձ�������������Ͳ��100mL����ƿ����ͷ�ιܣ����Ի���Ҫ��Ͳ���ձ���100mL����ƿ��
�ʴ�Ϊ����Ͳ���ձ���100mL����ƿ��
��4��A��ת����Һ��û��ϴ���ձ��ڱڣ���������ƿ��������������ʵ�����С��������ҺŨ��ƫ�ͣ���A���ϣ�
B���������ˮ���ݣ�����ǰ����ƿ��������ˮ����������ҺŨ����Ӱ�죬��B�����ϣ�
C������ʱ���ӣ�������Һ���ƫ��������ҺŨ��ƫ�ͣ���C���ϣ�
D����Һ���������������ʣ�Ũ����ϡ�ͣ��ų��������ȣ�ϡ�ͺ�δ�ָ���������ת�Ƶ�����ƿ�ж��ݣ�����������Һ�����С��������ҺŨ��ƫ�ߣ���D�����ϣ�
��ѡ��AC��
�ʴ�Ϊ5.4��1��2cm��
��1�����Ʋ�������ȡ��ϡ�͡���Һ��ϴ�ӡ����ݡ�ҡ�ȵȲ�����һ������Ͳ��ȡ���õ���ͷ�ιܣ����������ձ���ϡ�ͣ���ȴ��ת�Ƶ�100mL����ƿ�У����ò�����������ת����ϣ�����������ˮϴ���ձ���������2��3�β���ϴ��Һȫ��ת�Ƶ�����ƿ�У��ټ���������ˮ������ˮ��Һ�����̶���1��2cmʱ�����ý�ͷ�ιܵμӣ�ʹ��Һ�İ�Һ�����͵��������ƽ������ƿ�����������µߵ�ҡ�ȣ�
�ʴ�Ϊ��DACBEGF��
��2���ɣ�1�����������֪��ϡ�ͺ������ȴ�����£���ת�Ƶ�����ƿ�У�
�ʴ�Ϊ����ȴ�����£�
��3���ɣ�1�����������֪�������������ձ�������������Ͳ��100mL����ƿ����ͷ�ιܣ����Ի���Ҫ��Ͳ���ձ���100mL����ƿ��
�ʴ�Ϊ����Ͳ���ձ���100mL����ƿ��
��4��A��ת����Һ��û��ϴ���ձ��ڱڣ���������ƿ��������������ʵ�����С��������ҺŨ��ƫ�ͣ���A���ϣ�
B���������ˮ���ݣ�����ǰ����ƿ��������ˮ����������ҺŨ����Ӱ�죬��B�����ϣ�
C������ʱ���ӣ�������Һ���ƫ��������ҺŨ��ƫ�ͣ���C���ϣ�
D����Һ���������������ʣ�Ũ����ϡ�ͣ��ų��������ȣ�ϡ�ͺ�δ�ָ���������ת�Ƶ�����ƿ�ж��ݣ�����������Һ�����С��������ҺŨ��ƫ�ߣ���D�����ϣ�
��ѡ��AC��
�������⿼����һ�����ʵ���Ũ����Һ�����ƣ�ע���c=
��������ԭ����ע��Ũ�����ϡ�Ͳ�����
| n |
| V |

��ϰ��ϵ�д�
�����Ŀ
��18.4mol?L-1��Ũ��������4mol?L-1��������Һʱ������������������ҺŨ��ƫ�ߵ��ǣ�������
| A����ϡ�͵�������Һת��������ƿ��δϴ���ձ��Ͳ����� | B�����ձ��ڵ�ϡ����������ƿ��ת��ʱ�����������ʹ����ϡ���ὦ��ƿ�� | C���ý�ͷ�ιܼ�ˮʱ�������ӹ۲���Һ��Һ��������ƿ�̶����� | D���ý�ͷ�ι�������ƿ�м�ˮʱ��Һ�����������ƿ�̶ȣ���ʱ�����õιܽ�ƿ��Һ��������ʹ��Һ��Һ����̶����� |