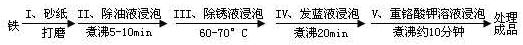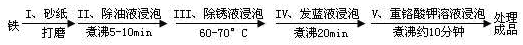��Ŀ����
�������������Ĺ��̿��������»�ѧ����ʽ��ʾ������˵����ȷ����
��3Fe+NaNO2+5NaOH�T3Na2FeO2+H2O+NH3��
��[x]Na2FeO2+[y]NaNO2+[z]H2O�T[m]Na2Fe2O4+[n]NH3��+[k]NaOH
��Na2FeO2+Na2Fe2O4+2H2O�TFe3O4+4NaOH
- A.��Ӧ�ٲ���������ԭ��Ӧ
- B.���������̲������κ���Ⱦ
- C.��Ӧ���У�ÿ��5.6gFe�μӷ�Ӧת��0.8mol����
- D.��Ӧ���е�z��m��k������5��3��7
D
������A����Ӧ����FeԪ�صĻ��ϼ����ߣ�NԪ�صĻ��ϼ۽��ͣ�
B����Ӧ�����ɰ��������д̼�����ζ��
C����Ӧ����FeԪ����0����Ϊ+2�ۣ�
D������ԭ���غ���������ѧ����������ֵ��
���A����Ӧ����FeԪ�صĻ��ϼ����ߣ�NԪ�صĻ��ϼ۽��ͣ���÷�Ӧ����������ԭ��Ӧ����A����
B���ɷ�Ӧ�٢ڿ�֪����Ӧ�����ɰ��������д̼�����ζ������Ⱦ��������B����
C����Ӧ����FeԪ����0����Ϊ+2�ۣ���ÿ��5.6gFe�μӷ�Ӧת��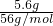 ����2-0��=0.2mol���ӣ���C����
����2-0��=0.2mol���ӣ���C����
D����ԭ���غ��֪��2x+y=2m+k��x=2m��y=n��2x+2y+z=4m+k����z��m��k������5��3��7����D��ȷ��
��ѡD��
���������⿼��������ԭ��Ӧ����ȷ��Ӧ��Ԫ�صĻ��ϼ۱仯��ԭ���غ㼴�ɽ����Ŀ�Ѷ��еȣ�
������A����Ӧ����FeԪ�صĻ��ϼ����ߣ�NԪ�صĻ��ϼ۽��ͣ�
B����Ӧ�����ɰ��������д̼�����ζ��
C����Ӧ����FeԪ����0����Ϊ+2�ۣ�
D������ԭ���غ���������ѧ����������ֵ��
���A����Ӧ����FeԪ�صĻ��ϼ����ߣ�NԪ�صĻ��ϼ۽��ͣ���÷�Ӧ����������ԭ��Ӧ����A����
B���ɷ�Ӧ�٢ڿ�֪����Ӧ�����ɰ��������д̼�����ζ������Ⱦ��������B����
C����Ӧ����FeԪ����0����Ϊ+2�ۣ���ÿ��5.6gFe�μӷ�Ӧת��
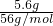 ����2-0��=0.2mol���ӣ���C����
����2-0��=0.2mol���ӣ���C����D����ԭ���غ��֪��2x+y=2m+k��x=2m��y=n��2x+2y+z=4m+k����z��m��k������5��3��7����D��ȷ��
��ѡD��
���������⿼��������ԭ��Ӧ����ȷ��Ӧ��Ԫ�صĻ��ϼ۱仯��ԭ���غ㼴�ɽ����Ŀ�Ѷ��еȣ�

��ϰ��ϵ�д�
 ��У����ϵ�д�
��У����ϵ�д�
�����Ŀ