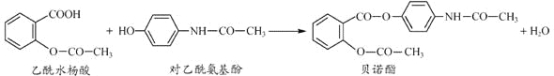
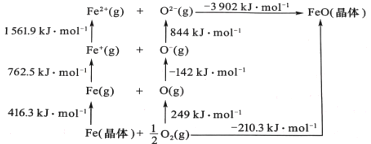��Ŀ����
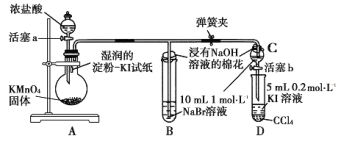
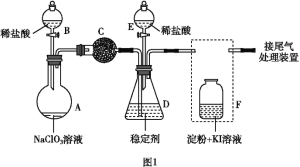
����Ŀ��ClO2(����ɫ������ˮ������)��һ�ָ�Ч���Ͷ�����������ij��ȤС��ͨ��ͼ1װ��(�г�װ����)�����Ʊ������պ��ͷŽ������о���
(1)����A��������_______________________��
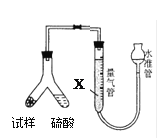
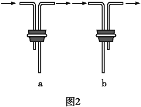
(2)��װF�е���ʱ��Ӧѡ��ͼ2�е�___________________��
(3)A�з�����Ӧ����ClO2��Cl2������������ͻ�ԭ�������ʵ���֮��Ϊ__________��
(4)�ر�B�Ļ�����ClO2��D�б��ȶ�����ȫ��������NaClO2����ʱF����Һ����ɫ���䣬��װ��C��������__________________��
(5)��֪������������NaClO2�ɷ�����Ӧ����NaCl���ͷų�ClO2���÷�Ӧ�����ӷ���ʽΪ____��
(6)ClO2�ܲ��ȶ������������ƣ�������ˮ���տɵõ�ClO2��Һ��Ϊ�ⶨ������Һ��ClO2��Ũ�ȣ�����������ʵ�飺
����1��ȷ��ȡClO2��Һ10.00mL��ϡ�ͳ�100.00mL��������ȡV0 mL�������뵽��ƿ�У�
����2����ϡ�������������pH��2.0������������KI���壬����Ƭ�̣�
����3������ָʾ������c mol��L1 Na2S2O3��Һ�ζ����յ㡣�ظ�2�Σ��������Na2S2O3��Һƽ��ֵΪV1 mL��
(��֪2ClO2+10I+8H+=2Cl+5I2+4H2O��2Na2S2O3+I2![]() Na2S4O6+2NaI)
Na2S4O6+2NaI)
�����ClO2��Ũ��Ϊ______g/L(�ú���ĸ�Ĵ���ʽ��ʾ������������)��
���𰸡�Բ����ƿ b 1��2 ����Cl2 4H++5ClO2-=Cl+4ClO2��+2H2O 135cV1/V0
��������
����ʵ��װ��ͼ��֪��A��B���Ʊ������Ͷ������ȵģ�C���������������ģ�D���������ն������ȵģ�E�����������ͷŶ������ȵģ�F�ǿ���֤β�����Ƿ���������
(1)����������������֪����A��Բ����ƿ��
(2)Fװ��Ӧ��Cl2��KI��Ӧ������Ҫ����β������װ�ã�����Ӧ���ܽ������̹ܳ�������ѡb��
(3)�����ƺ�ϡ�����ϲ���Cl2��ClO2��NaClO3����Ϊ+5�۱���ԭ��ClO2��+4�ۣ���������Ϊ-1�ۣ���������0���ȣ�ת�Ƶ�����Ϊ2������NaClO3��ClO2ǰ��ѧ������Ϊ2��Cl2ǰ��ѧ������Ϊ1����Ӧ��ѧ����ʽ��2NaClO3+4HCl=2ClO2��+Cl2��+2NaCl+2H2O������������Cl2�ͻ�ԭ����ClO2���ʵ���֮��Ϊ1��2��
(4)Fװ���з���Cl2+2KI=2KCl+I2ʱ���������۱�������F����Һ����ɫ���䣬��װ��C������������Cl2��
(5)������������NaClO2�ɷ�����Ӧ����NaCl���ͷų�ClO2������Ԫ���غ��֪Ӧ����ˮ���ɣ��÷�Ӧ�����ӷ���ʽΪ4H++5ClO2-=Cl+4ClO2��+2H2O��
(6)��ԭClO2��Һ��Ũ��Ϊx mol/L������ݵζ������еķ�Ӧ����ʽ��֪2ClO2��5I2��10Na2S2O3������![]() �����x=
�����x=![]() �����㵥λ��
�����㵥λ��![]() ��
��

 ����С״Ԫ��������������ϵ�д�
����С״Ԫ��������������ϵ�д�����Ŀ����ʽ������(NiOOH)�����������ص��������ϣ����÷�����������Ҫ��Ni��Al������Cr��FeS ��)���Ʊ����乤���������£�

�ش��������⣺
(1)�����ݳ�����ʱ��������Ӧ�����ӷ�Ӧ����ʽΪ_________________________;
(2)���ܽ⡱ʱ�ų�������Ϊ_______________ (�ѧʽ);
(3)��֪�������½������ӿ�ʼ��������ȫ������pH���±���
��ʼ������pH | ��ȫ������pH | |
Ni2+ | 6.2 | 8.6 |
Fe2+ | 7.6 | 9.1 |
Fe3+ | 2.3 | 3.3 |
Cr3+ | 4.5 | 5.6 |
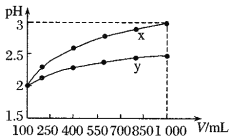
����pH 1��ʱ����ҺpH��ΧΪ______________________��
(4)�ڿ����м���Ni(OH)2�ɵ�NiOOH,��д���˷�Ӧ�Ļ�ѧ����ʽ_____________;

(5)����������Һ���ж��ִ�����ʽ, CrO42����Cr2O72������Һ�п��ת���������£���ʼŨ��Ϊ1.0mol/L��Na2CrO4��Һ��c(Cr2O72��)��c(H+)�ı仯��ͼ��ʾ�������ӷ���ʽ��ʾNa2CrO4��Һ�е�ת����Ӧ________________������A�����ݼ������ת����Ӧ��ƽ�ⳣ��Ϊ______________,�¶����ߣ���Һ��CrO42����ƽ��ת���ʼ�С����÷�Ӧ�ġ�H____0���>������<����=������