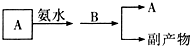��Ŀ����
������A��B����ѧ���������ʣ����������ӿɴ��±���ѡ��
��1����A��B��ˮ��Һ��Ϊ��ɫ��B��ˮ��Һ�ʼ��ԣ��һ�Ϻ�ֻ����������ϡ����İ�ɫ��������ʹ��ɫʯ����ֽ���������壮
��B�Ļ�ѧʽΪ
��A��B��Һ��Ϻ���ȳ����Է�Ӧ�����ӷ���ʽΪ
��2����A��ˮ��Һ��dz��ɫ��B��ˮ��Һ��ɫ������ɫ��ӦΪ��ɫ����A��ˮ��Һ�м���ϡ���������������ټ���B����Һ��ƣ���A��B��ˮ��Һ����������Ա仯�����AΪ
�ھ�����������������Һ��Ƶ�ԭ������������֣���
������һ������֤��������Һ��Ƶ�ԭ��
��������Һ���ԭ����������Ƴ�ԭ��أ�����1.8mol������a����b����b���ĵ缫��ӦʽΪ
| ������ | K+ Na+ Fe2+ Ba2+ NH4+ |
| ������ | OH- NO3- I- HCO3- AlO2- HSO4- |
��B�Ļ�ѧʽΪ
Ba��OH��2
Ba��OH��2
����A��B��Һ��Ϻ���ȳ����Է�Ӧ�����ӷ���ʽΪ
H++SO42-+NH4++Ba2++2OH-
BaSO4��+NH3��+2H2O
| ||
H++SO42-+NH4++Ba2++2OH-
BaSO4��+NH3��+2H2O
| ||
��2����A��ˮ��Һ��dz��ɫ��B��ˮ��Һ��ɫ������ɫ��ӦΪ��ɫ����A��ˮ��Һ�м���ϡ���������������ټ���B����Һ��ƣ���A��B��ˮ��Һ����������Ա仯�����AΪ
FeI2
FeI2
��BΪNaNO3
NaNO3
���ھ�����������������Һ��Ƶ�ԭ������������֣���
����I-��������I2ʹ��Һ�ʻ�ɫ
����I-��������I2ʹ��Һ�ʻ�ɫ
��I-��Fe2+��������ʹ��Һ�ʻ�ɫ
I-��Fe2+��������ʹ��Һ�ʻ�ɫ
��������һ������֤��������Һ��Ƶ�ԭ��
ȡ���������Һ���Թ��У��μӼ���KSCN��Һ�������������
ȡ���������Һ���Թ��У��μӼ���KSCN��Һ�������������
����������Һ���ԭ����������Ƴ�ԭ��أ�����1.8mol������a����b����b���ĵ缫��ӦʽΪ
NO3-+4H++3e-�TNO��+2H2O
NO3-+4H++3e-�TNO��+2H2O
����a��b��Ϊʯī�缫��b����ԭ
��ԭ
�����������ԭ������������ʵ�����0.6mol
0.6mol
����������1���ٸ��ݡ���Ϻ�ֻ����������ϡ����İ�ɫ��������ʹ��ɫʯ����ֽ���������塱�ƶ�A��B�к�������ӡ������ӡ�����������Ӻ����������ӣ��ٸ���B��ˮ��Һ�ʼ����ж�A��B�����ƣ�
���������������������Ӧ���ȷ�Ӧ�������ᱵ��������ˮ���ݴ�д����Ӧ�����ӷ���ʽ��
��2������Һ��dz��ɫ����Һ�к����������ӣ���ɫ��ӦΪ��ɫ����Һ�к��������ӣ���A��ˮ��Һ�м���ϡ���������������ټ���B����Һ��ƣ�˵�������������·�����������ԭ��Ӧ��A��B����Һһ��������������ӣ���A��ˮ��Һ�м���ϡ��������������˵��A�в��������������ӣ��ٸ������ӹ����ж�A��B����ɣ�
�ڵ����Ӻ��������ӱ����������ɵĵⵥ�ʡ������Ӷ��ܹ�ʹ��Һ��ʾ��ɫ�������ӻ�ԭ��ǿ���������ӣ�
�ۿ���ͨ�����з�Ӧ�����Һ���Ƿ���������ӣ��ж�������Һ��Ƶ�ԭ��
�ܵ�����a����b��a������������Ӧ��b��������ԭ��Ӧ��aΪ������bΪ������b��������ԭ��Ӧ�����ݵ����غ�������ԭ��������ʵ�����
���������������������Ӧ���ȷ�Ӧ�������ᱵ��������ˮ���ݴ�д����Ӧ�����ӷ���ʽ��
��2������Һ��dz��ɫ����Һ�к����������ӣ���ɫ��ӦΪ��ɫ����Һ�к��������ӣ���A��ˮ��Һ�м���ϡ���������������ټ���B����Һ��ƣ�˵�������������·�����������ԭ��Ӧ��A��B����Һһ��������������ӣ���A��ˮ��Һ�м���ϡ��������������˵��A�в��������������ӣ��ٸ������ӹ����ж�A��B����ɣ�
�ڵ����Ӻ��������ӱ����������ɵĵⵥ�ʡ������Ӷ��ܹ�ʹ��Һ��ʾ��ɫ�������ӻ�ԭ��ǿ���������ӣ�
�ۿ���ͨ�����з�Ӧ�����Һ���Ƿ���������ӣ��ж�������Һ��Ƶ�ԭ��
�ܵ�����a����b��a������������Ӧ��b��������ԭ��Ӧ��aΪ������bΪ������b��������ԭ��Ӧ�����ݵ����غ�������ԭ��������ʵ�����
����⣺��1����A��B��ˮ��Һ��Ϊ��ɫ��B��ˮ��Һ�ʼ��ԣ��һ�Ϻ�ֻ����������ϡ����İ�ɫ��������ʹ��ɫʯ����ֽ���������壬����Ϊ���ᱵ������Ϊ������˵��A��B�к�����������ӡ������ӡ�����Ӻ����������ӣ�Bˮ��Һ��ʾ���ԣ�B�к������������ӣ��������ӹ��棬BΪBa��OH��2����AΪ������泥�
�ʴ�Ϊ��Ba��OH��2��
��������������������������ʵ���1��1��Ӧ����Ӧ�����Һ��ʾ���ԣ���Ӧ�����ӷ���ʽΪ��H++SO42-+NH4++Ba2++2OH-
BaSO4��+NH3��+2H2O��
�ʴ�Ϊ��H++SO42-+NH4++Ba2++2OH-
BaSO4��+NH3��+2H2O��
��2����A��ˮ��Һ��dz��ɫ����A��Һ�к���Fe2+��B��ˮ��Һ��ɫ������ɫ��ӦΪ��ɫ����B��Һ�к���Na+����A��ˮ��Һ�м���ϡ���������������ټ���B����Һ��ƣ�˵��A��B��һ��������������ӣ�������A��ˮ��Һ�м���ϡ��������������˵�������������B�У���BΪNaNO3���ܹ������������γɿ����Ե�������I-��HSO4-�����ڡ�A��B��ˮ��Һ����������Ա仯��˵��A��һ�����������ӣ�����AΪFeI2��
�ʴ�Ϊ��FeI2��NaNO3��
��A��Һ�е��������Ӻ͵����Ӷ����л�ԭ�ԣ��ҵ����ӻ�ԭ��ǿ���������ӣ�����������Ӳ��㣬��Һ��Ƶ�ԭ������ӱ������ɵⵥ��ʹ��Һ�ʻ�ɫ�����������������������������Ӻ���ʣ�࣬�ܹ����������������ӣ�������Һ�ʻ�ɫI-��Fe2+����������
�ʴ�Ϊ������I-��������I2ʹ��Һ�ʻ�ɫ��I-��Fe2+��������ʹ��Һ�ʻ�ɫ��
������Ӧ�����Һ�д��������ӣ�֤�����������������Ϊ��ȡ���������Һ���Թ��У��μӼ���KSCN��Һ�������������������������
�ʴ�Ϊ��ȡ���������Һ���Թ��У��μӼ���KSCN��Һ�������������������������ɣ���
����1.8mol������a����b����b��������ԭ��Ӧ�������ĵ缫��ӦΪ��NO3-+4H++3e-�TNO��+2H2O��b����ԭ����Ϊһ�����������ݵ����غ㣬���ɵ�һ�����������ʵ���Ϊ��
=0.6mol��
�ʴ�Ϊ��NO3-+4H++3e-�TNO��+2H2O����ԭ��0.6mol��
�ʴ�Ϊ��Ba��OH��2��
��������������������������ʵ���1��1��Ӧ����Ӧ�����Һ��ʾ���ԣ���Ӧ�����ӷ���ʽΪ��H++SO42-+NH4++Ba2++2OH-
| ||
�ʴ�Ϊ��H++SO42-+NH4++Ba2++2OH-
| ||
��2����A��ˮ��Һ��dz��ɫ����A��Һ�к���Fe2+��B��ˮ��Һ��ɫ������ɫ��ӦΪ��ɫ����B��Һ�к���Na+����A��ˮ��Һ�м���ϡ���������������ټ���B����Һ��ƣ�˵��A��B��һ��������������ӣ�������A��ˮ��Һ�м���ϡ��������������˵�������������B�У���BΪNaNO3���ܹ������������γɿ����Ե�������I-��HSO4-�����ڡ�A��B��ˮ��Һ����������Ա仯��˵��A��һ�����������ӣ�����AΪFeI2��
�ʴ�Ϊ��FeI2��NaNO3��
��A��Һ�е��������Ӻ͵����Ӷ����л�ԭ�ԣ��ҵ����ӻ�ԭ��ǿ���������ӣ�����������Ӳ��㣬��Һ��Ƶ�ԭ������ӱ������ɵⵥ��ʹ��Һ�ʻ�ɫ�����������������������������Ӻ���ʣ�࣬�ܹ����������������ӣ�������Һ�ʻ�ɫI-��Fe2+����������
�ʴ�Ϊ������I-��������I2ʹ��Һ�ʻ�ɫ��I-��Fe2+��������ʹ��Һ�ʻ�ɫ��
������Ӧ�����Һ�д��������ӣ�֤�����������������Ϊ��ȡ���������Һ���Թ��У��μӼ���KSCN��Һ�������������������������
�ʴ�Ϊ��ȡ���������Һ���Թ��У��μӼ���KSCN��Һ�������������������������ɣ���
����1.8mol������a����b����b��������ԭ��Ӧ�������ĵ缫��ӦΪ��NO3-+4H++3e-�TNO��+2H2O��b����ԭ����Ϊһ�����������ݵ����غ㣬���ɵ�һ�����������ʵ���Ϊ��
| 1.8mol |
| 5-2 |
�ʴ�Ϊ��NO3-+4H++3e-�TNO��+2H2O����ԭ��0.6mol��
���������⿼���˳������ӵļ��顢δ֪����ƶϡ����ӷ���ʽ��д��ԭ���ԭ�����缫��Ӧʽ��д��֪ʶ����Ŀ�Ѷ��еȣ������漰�������ϴ�֪ʶ��϶࣬��ֿ�����ѧ������ѧ֪ʶ�����������

��ϰ��ϵ�д�
 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�
�����Ŀ
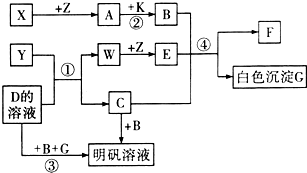
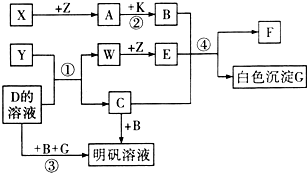 ��ѧ��ѧ�г�������������ͼ��ʾ��ת����ϵ����Ӧ������ȥ����X��Y��Z��W�ǵ��ʣ������Ϊ�����A��W��Z�����³���̬����A��һ�ִ�����Ⱦ�B��һ�ֳ��õ��ᣮ
��ѧ��ѧ�г�������������ͼ��ʾ��ת����ϵ����Ӧ������ȥ����X��Y��Z��W�ǵ��ʣ������Ϊ�����A��W��Z�����³���̬����A��һ�ִ�����Ⱦ�B��һ�ֳ��õ��ᣮ
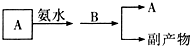
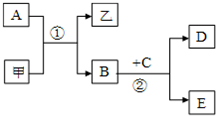 ͼ��ʾ��ת����ϵ�У��ס��Ҽ�A��B��C��D��E��Ϊ��ѧ�����Ļ�ѧ���ʣ����мס���Ϊ�����A��B��DΪ���ʣ�
ͼ��ʾ��ת����ϵ�У��ס��Ҽ�A��B��C��D��E��Ϊ��ѧ�����Ļ�ѧ���ʣ����мס���Ϊ�����A��B��DΪ���ʣ�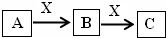 A��B��C��X����ѧ��ѧ�г��������ʣ�����֮���ת����ϵ��ͼ��ʾ�����ֲ�������ȥ������ش��������⣺
A��B��C��X����ѧ��ѧ�г��������ʣ�����֮���ת����ϵ��ͼ��ʾ�����ֲ�������ȥ������ش��������⣺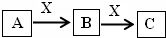 A��B��C��X����ѧ��ѧ�г��������ʣ�����֮���ת����ϵ����ͼ��ʾ�����ֲ�������ȥ������ش��������⣺
A��B��C��X����ѧ��ѧ�г��������ʣ�����֮���ת����ϵ����ͼ��ʾ�����ֲ�������ȥ������ش��������⣺