��Ŀ����
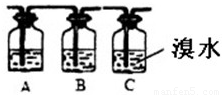
 ij��ѧ��ȤС����ȡ��������ϩ���ʵ�̽��ʵ�飬�Ƶõ���ϩ�п��ܺ���SO2��CO2�����壬���Ƶõ���ϩͨ����ˮʱ���ɹ۲쵽�Թ�����ˮ��ɫ��
ij��ѧ��ȤС����ȡ��������ϩ���ʵ�̽��ʵ�飬�Ƶõ���ϩ�п��ܺ���SO2��CO2�����壬���Ƶõ���ϩͨ����ˮʱ���ɹ۲쵽�Թ�����ˮ��ɫ����1����ͬѧ��Ϊ�����ǵ���Ӧ�ĸ����ԣ�ʹ��ˮ��ɫ�����ʲ�һ������ϩ�������ǣ�
SO2���л�ԭ�ԣ�Ҳ������ˮ������Ӧʹ��ˮ��ɫ��
SO2���л�ԭ�ԣ�Ҳ������ˮ������Ӧʹ��ˮ��ɫ��
����2����ͬѧ�������ͼʵ�飬֤����ϩ����ʹ��ˮ��ɫ����A�е��Լ�ӦΪ
NaOH��Һ
NaOH��Һ
����������ȥ��������е�SO2��CO2
��ȥ��������е�SO2��CO2
��B�е��Լ�ӦΪƷ����Һ��
Ʒ����Һ��
������������SO2�Ƿ����
����SO2�Ƿ����
����3����ͬѧ��ȷ��д����ˮ����ϩ��Ӧ�Ļ�ѧ����ʽΪ��
CH2=CH2+Br2��CH2BrCH2Br
CH2=CH2+Br2��CH2BrCH2Br
����������1������SO2���л�ԭ�ԣ�Ҳ������ˮ��Ӧʹ��ˮ��ɫ��
��2������Aװ����Ϊ�˳�ȥSO2��CO2��Bװ����Ϊ�˼���SO2�Ƿ������
��3����ˮ�е�������ϩ�����ӳɷ�Ӧ��
��2������Aװ����Ϊ�˳�ȥSO2��CO2��Bװ����Ϊ�˼���SO2�Ƿ������
��3����ˮ�е�������ϩ�����ӳɷ�Ӧ��
����⣺��1����SO2���л�ԭ�ԣ�Ҳ������ˮ������Ӧʹ��ˮ��ɫ���ʴ�Ϊ��SO2���л�ԭ�ԣ�Ҳ������ˮ������Ӧʹ��ˮ��ɫ��
��2����Aװ����Ϊ�˳�ȥSO2������NaOH��Һ��Bװ����Ϊ�˼���SO2�Ƿ����������Ʒ����Һ���ʴ�Ϊ��NaOH��Һ����ȥ��������е�SO2��CO2��Ʒ����Һ������SO2�Ƿ������
��3����ˮ�е�������ϩ�����ӳɷ�Ӧ����CH2=CH2+Br2��CH2BrCH2Br���ʴ�Ϊ��CH2=CH2+Br2��CH2BrCH2Br��
��2����Aװ����Ϊ�˳�ȥSO2������NaOH��Һ��Bװ����Ϊ�˼���SO2�Ƿ����������Ʒ����Һ���ʴ�Ϊ��NaOH��Һ����ȥ��������е�SO2��CO2��Ʒ����Һ������SO2�Ƿ������
��3����ˮ�е�������ϩ�����ӳɷ�Ӧ����CH2=CH2+Br2��CH2BrCH2Br���ʴ�Ϊ��CH2=CH2+Br2��CH2BrCH2Br��
������������ʵ�鿼������ϩ�Ļ�ѧ���ʣ��ѶȲ���������ѧ���������⡢��������������

��ϰ��ϵ�д�
 ��������һ���þ�ϵ�д�
��������һ���þ�ϵ�д� Сѧ��10����Ӧ����ϵ�д�
Сѧ��10����Ӧ����ϵ�д�
�����Ŀ
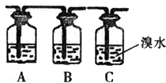 ij��ѧ��ȤС����ȡ��������ϩ���ʵ�̽��ʵ�飬�Ƶõ���ϩ�п��ܺ���SO2��CO2�����壬���Ƶõ���ϩͨ����ˮʱ���ɹ۲쵽�Թ�����ˮ��ɫ��
ij��ѧ��ȤС����ȡ��������ϩ���ʵ�̽��ʵ�飬�Ƶõ���ϩ�п��ܺ���SO2��CO2�����壬���Ƶõ���ϩͨ����ˮʱ���ɹ۲쵽�Թ�����ˮ��ɫ��