��Ŀ����
ij��ѧ��ȤС��ͬѧչ����Ư���������ƣ�NaClO2�����о���
ʵ�����ȡNaClO2����
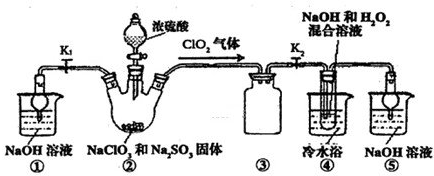
��֪��NaClO2������Һ���¶ȵ���38��ʱ�����ľ�����NaClO2?3H2O������38��ʱ�����ľ�����NaClO2������60��ʱNaClO2�ֽ��NaClO3��NaCl��������ͼ��ʾװ�ý���ʵ�飮
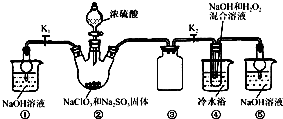
��1��װ�â۵�������
��2��װ�â��в���ClO2�Ļ�ѧ����ʽΪ
��3����װ�âܷ�Ӧ�����Һ���NaClO2����IJ�������Ϊ��
�ټ�ѹ��55�������ᾧ���ڳ��ȹ��ˣ���
ʵ��ⶨij����������Ʒ�Ĵ��ȣ�
�������ʵ�鷽����������ʵ�飺
��ȷ��ȡ��������������Ʒm g���ձ��У�������������ˮ�����ĵ⻯�ؾ��壬�ٵ���������ϡ���ᣬ��ַ�Ӧ����֪��Cl
+4I-+4H+�T2H2O+2I2+Cl-���������û��Һ���250mL������Һ��
����ȡ25.00mL������Һ����ƿ�У��Ӽ��ε�����Һ����c mol?L-1 Na2S2O3��Һ�ζ������ζ��յ㣮�ظ�2�Σ����ƽ��ֵΪV mL����֪��I2+2S2
�T2I-+S4
��
��4���ﵽ�ζ��յ�ʱ������Ϊ
��5������Ʒ��NaClO2����������Ϊ
%
%���ú�m��c��V�Ĵ���ʽ��ʾ����
��6���ڵζ�������ȷ���������£���ʵ���ý��ƫ�ߣ�ԭ�������ӷ���ʽ��ʾΪ
ʵ�����ȡNaClO2����
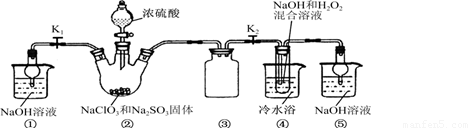
��֪��NaClO2������Һ���¶ȵ���38��ʱ�����ľ�����NaClO2?3H2O������38��ʱ�����ľ�����NaClO2������60��ʱNaClO2�ֽ��NaClO3��NaCl��������ͼ��ʾװ�ý���ʵ�飮

��1��װ�â۵�������
��ֹ����
��ֹ����
����2��װ�â��в���ClO2�Ļ�ѧ����ʽΪ
2NaClO3+Na2SO3+H2SO4=2ClO2��+2Na2SO4+H2O
2NaClO3+Na2SO3+H2SO4=2ClO2��+2Na2SO4+H2O
��װ�â����Ʊ�NaClO2�Ļ�ѧ����ʽΪ2NaOH+2ClO2+H2O2=2NaClO2+2H2O+O2
2NaOH+2ClO2+H2O2=2NaClO2+2H2O+O2
����3����װ�âܷ�Ӧ�����Һ���NaClO2����IJ�������Ϊ��
�ټ�ѹ��55�������ᾧ���ڳ��ȹ��ˣ���
��38�桫60����ˮϴ��
��38�桫60����ˮϴ��
���ܵ���60�����õ���Ʒ��ʵ��ⶨij����������Ʒ�Ĵ��ȣ�
�������ʵ�鷽����������ʵ�飺
��ȷ��ȡ��������������Ʒm g���ձ��У�������������ˮ�����ĵ⻯�ؾ��壬�ٵ���������ϡ���ᣬ��ַ�Ӧ����֪��Cl
| O | - 2 |
����ȡ25.00mL������Һ����ƿ�У��Ӽ��ε�����Һ����c mol?L-1 Na2S2O3��Һ�ζ������ζ��յ㣮�ظ�2�Σ����ƽ��ֵΪV mL����֪��I2+2S2
| O | - 2 |
| O | 2- 6 |
��4���ﵽ�ζ��յ�ʱ������Ϊ
�μ����һ��Na2S2O3��Һʱ��Һ����ɫ��Ϊ��ɫ�Ұ�����ڲ���ɫ��˵������ζ��յ�
�μ����һ��Na2S2O3��Һʱ��Һ����ɫ��Ϊ��ɫ�Ұ�����ڲ���ɫ��˵������ζ��յ�
����5������Ʒ��NaClO2����������Ϊ
| 90.5cV |
| 4m |
| 90.5cV |
| 4m |
��6���ڵζ�������ȷ���������£���ʵ���ý��ƫ�ߣ�ԭ�������ӷ���ʽ��ʾΪ
4I-+O2+4H+=2I2+2H2O
4I-+O2+4H+=2I2+2H2O
����������1��װ�â������巴Ӧ��װ����ѹǿ���ͣ�װ�â۷�ֹ������
��2���������ƾ��л�ԭ�ԣ��ڷ�Ӧ������ԭ����
װ�âܷ�Ӧ�����Һ���NaClO2���壬��װ�â�������NaClO2��ClԪ�صĻ��ϼ۽��ͣ�˫��ˮӦ���ֻ�ԭ�ԣ����������ɣ����ԭ���غ��֪������ˮ���ɣ���ƽ��д����ʽ��
��3������Һ����ȡ���壬һ����������ᾧ�����ˡ�ϴ�ӡ�����ķ�����ע���¶ȿ��ƣ�
��4����Ӧ����ʱ���ⷴӦ��ȫ���μ����һ��Һ��ʱ��Һ����ɫ��Ϊ��ɫ�Ұ�����ڲ���ɫ��˵������ζ��յ㣻
��5�����ݹ�ϵʽNaClO2��2I2��4S2O32-���м��㣻
��6��ʵ���ý��ƫ�ߣ�˵���ζ����ĵ�Na2S2O3��Һ���ƫ�ߣ���Һ�е�ĺ���ƫ�ߣ�Ӧ�����ɵĵ����ӱ���������Ϊ�����£�
��2���������ƾ��л�ԭ�ԣ��ڷ�Ӧ������ԭ����
װ�âܷ�Ӧ�����Һ���NaClO2���壬��װ�â�������NaClO2��ClԪ�صĻ��ϼ۽��ͣ�˫��ˮӦ���ֻ�ԭ�ԣ����������ɣ����ԭ���غ��֪������ˮ���ɣ���ƽ��д����ʽ��
��3������Һ����ȡ���壬һ����������ᾧ�����ˡ�ϴ�ӡ�����ķ�����ע���¶ȿ��ƣ�
��4����Ӧ����ʱ���ⷴӦ��ȫ���μ����һ��Һ��ʱ��Һ����ɫ��Ϊ��ɫ�Ұ�����ڲ���ɫ��˵������ζ��յ㣻
��5�����ݹ�ϵʽNaClO2��2I2��4S2O32-���м��㣻
��6��ʵ���ý��ƫ�ߣ�˵���ζ����ĵ�Na2S2O3��Һ���ƫ�ߣ���Һ�е�ĺ���ƫ�ߣ�Ӧ�����ɵĵ����ӱ���������Ϊ�����£�
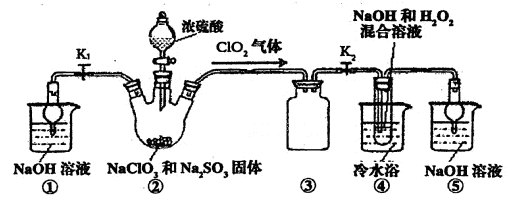
����⣺��1��װ�â��ǰ�ȫƿ�ܷ�ֹ������
�ʴ�Ϊ����ֹ������
��2���������ƾ��л�ԭ�ԣ��ڷ�Ӧ������ԭ����װ�â��в���ClO2�ķ�Ӧ����������������Һ��������������Ϊ�����ƣ���������ԭΪ�������ȣ���Ӧ�Ļ�ѧ����ʽΪ��2NaClO3+Na2SO3+H2SO4=2ClO2��+2Na2SO4+H2O��װ�âܷ�Ӧ�����Һ���NaClO2���壬��װ�â�������NaClO2��ClԪ�صĻ��ϼ۽��ͣ�˫��ˮӦ���ֻ�ԭ�ԣ����������ɣ����ԭ���غ��֪������ˮ���ɣ���ƽ��ʽΪ��2NaOH+2ClO2+H2O2=2NaClO2+2H2O+O2��
�ʴ�Ϊ��2NaClO3+Na2SO3+H2SO4=2ClO2��+2Na2SO4+H2O��Na2SO3��2NaOH+2ClO2+H2O2=2NaClO2+2H2O+O2��
��3������Һ����ȡ���壬һ����������ᾧ�����ˡ�ϴ�ӡ�����ķ�����Ϊ��ֹ��������NaClO2?3H2O��Ӧ���ȹ��ˣ�����Ŀ��Ϣ��֪��Ӧ�����¶�38�桫60�����ϴ�ӣ�����60����
�ʴ�Ϊ����38�桫60����ˮϴ�ӣ�
��4���������۱���ɫ����Ӧ����ʱ���ⷴӦ��ȫ���μ����һ��Na2S2O3��Һʱ��Һ����ɫ��Ϊ��ɫ�Ұ�����ڲ���ɫ��˵������ζ��յ㣬
�ʴ�Ϊ���μ����һ��Na2S2O3��Һʱ��Һ����ɫ��Ϊ��ɫ�Ұ�����ڲ���ɫ��˵������ζ��յ㣻
��5������Ʒ��NaClO2����������Ϊa����
NaClO2��2I2��4S2O32-
90.5g 4mol
mag c mol?L-1��V��10-3L��
��
����90.5g��mag=4mol��c mol?L-1��V��10-3L��
��
���a=
%=
%��
�ʴ�Ϊ��
%��
��6��ʵ���ý��ƫ�ߣ�˵���ζ����ĵ�Na2S2O3��Һ���ƫ�ߣ���Һ�е�ĺ���ƫ�ߣ�Ӧ�����ɵĵ����ӱ���������Ϊ�⣬ͬʱ����ˮ����Ӧ���ӷ���ʽΪ4I-+O2+4H+=2I2+2H2O��
�ʴ�Ϊ��4I-+O2+4H+=2I2+2H2O��
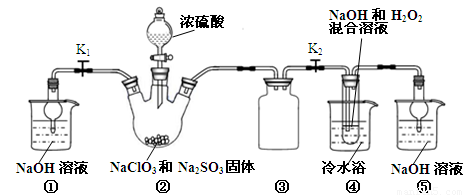
�ʴ�Ϊ����ֹ������
��2���������ƾ��л�ԭ�ԣ��ڷ�Ӧ������ԭ����װ�â��в���ClO2�ķ�Ӧ����������������Һ��������������Ϊ�����ƣ���������ԭΪ�������ȣ���Ӧ�Ļ�ѧ����ʽΪ��2NaClO3+Na2SO3+H2SO4=2ClO2��+2Na2SO4+H2O��װ�âܷ�Ӧ�����Һ���NaClO2���壬��װ�â�������NaClO2��ClԪ�صĻ��ϼ۽��ͣ�˫��ˮӦ���ֻ�ԭ�ԣ����������ɣ����ԭ���غ��֪������ˮ���ɣ���ƽ��ʽΪ��2NaOH+2ClO2+H2O2=2NaClO2+2H2O+O2��
�ʴ�Ϊ��2NaClO3+Na2SO3+H2SO4=2ClO2��+2Na2SO4+H2O��Na2SO3��2NaOH+2ClO2+H2O2=2NaClO2+2H2O+O2��
��3������Һ����ȡ���壬һ����������ᾧ�����ˡ�ϴ�ӡ�����ķ�����Ϊ��ֹ��������NaClO2?3H2O��Ӧ���ȹ��ˣ�����Ŀ��Ϣ��֪��Ӧ�����¶�38�桫60�����ϴ�ӣ�����60����
�ʴ�Ϊ����38�桫60����ˮϴ�ӣ�
��4���������۱���ɫ����Ӧ����ʱ���ⷴӦ��ȫ���μ����һ��Na2S2O3��Һʱ��Һ����ɫ��Ϊ��ɫ�Ұ�����ڲ���ɫ��˵������ζ��յ㣬
�ʴ�Ϊ���μ����һ��Na2S2O3��Һʱ��Һ����ɫ��Ϊ��ɫ�Ұ�����ڲ���ɫ��˵������ζ��յ㣻
��5������Ʒ��NaClO2����������Ϊa����
NaClO2��2I2��4S2O32-
90.5g 4mol
mag c mol?L-1��V��10-3L��
| 250ml |
| 25ml |
����90.5g��mag=4mol��c mol?L-1��V��10-3L��
| 250ml |
| 25ml |
���a=
| 90.5cV |
| 4m |
| 22.625cV |
| m |
�ʴ�Ϊ��
| 90.5cV |
| 4m |
��6��ʵ���ý��ƫ�ߣ�˵���ζ����ĵ�Na2S2O3��Һ���ƫ�ߣ���Һ�е�ĺ���ƫ�ߣ�Ӧ�����ɵĵ����ӱ���������Ϊ�⣬ͬʱ����ˮ����Ӧ���ӷ���ʽΪ4I-+O2+4H+=2I2+2H2O��
�ʴ�Ϊ��4I-+O2+4H+=2I2+2H2O��
���������⿼�����ʵ��Ʊ�������Ϣ�����á���װ�õ����⡢������ԭ��Ӧ�ζ��ȣ�����ԭ���ǽ���Ĺؼ���ͬʱ����ѧ���������⡢���������������ѶȽϴ�ע��������ԭ��Ӧ�ζ������ù�ϵʽ���еļ��㣮

��ϰ��ϵ�д�
�����Ŀ