��Ŀ����
�����������ķ�չ���������Nox���ѳ�Ϊ�����е���Ҫ��̬��Ⱦ��֮һ��
��1�������ٷɻ��ŷŵ�β����ƽ������NO����Ҫ��Դ�������ƻ�������Ļ���Ϊ��
��O3
O+O2
��NO��NO2+O2
��NO2+O��NO+O2
������Ӧ���ܷ�ӦʽΪ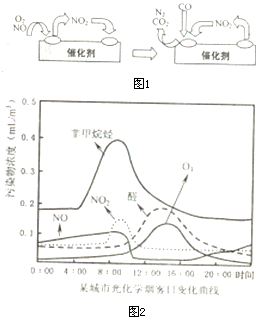
��2������β���к���NOx��CO��̼�⻯����ȣ�
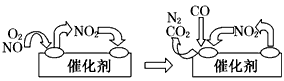
��β������װ����װ�к�Pd�ȹ���Ԫ�صĴ����������ڴ�������������������õĻ�����ͼ1��ʾ��β�����������з������ܷ�Ӧ��ѧ����ʽ��
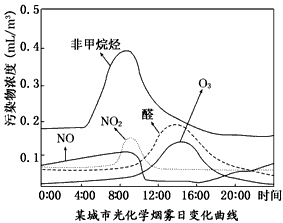
��NOx��̼�⻯���������������·����⻯ѧ��Ӧ����������Ⱦ�������Ϊ���⻯ѧ����������ѧ�߶�ij���У����й⻯ѧ�����ı仯������вⶨ��ʵ������ͼ2����ͼ���֪��������Ⱦ����
�۲ⶨ����β����Nox�ķ���֮һ����3%��H2O2��Һ����β���е�NOx����HNO3������NaOH����Һ�ζ�HNO3����Ҫ�ⶨ��״����β����NOx����������������������
A������β��������� B�����ֵ�������������
C��NaOH����Һ�����ʵ���Ũ�� D��������NaOH����Һ�����
E���ӵζ���ʼ��ָʾ����ɫ�����ʱ��
��3�����Ṥҵβ���е�NOx���ô�����Һ���գ��йصĻ�ѧ��ӦΪ��
2NO2+Na2O=NaNO3+CO2
NO+NO2+Na2CO3=2NaNO3+CO2
����0.5L 2mol/L�Ĵ�����Һǡ������һ�������Ṥҵβ���е�NOx������Һ��������44g����C02ȫ���ų�������NO2��NO�������Ϊ
��1�������ٷɻ��ŷŵ�β����ƽ������NO����Ҫ��Դ�������ƻ�������Ļ���Ϊ��
��O3
| �� |
��NO��NO2+O2
��NO2+O��NO+O2
������Ӧ���ܷ�ӦʽΪ
2O3
3O2
| ||
2O3
3O2
������
| ||
NO
NO
�Ǵ�����
��2������β���к���NOx��CO��̼�⻯����ȣ�
��β������װ����װ�к�Pd�ȹ���Ԫ�صĴ����������ڴ�������������������õĻ�����ͼ1��ʾ��β�����������з������ܷ�Ӧ��ѧ����ʽ��
2NO+O2+4CO
4CO2+N2
| ||
2NO+O2+4CO
4CO2+N2
��
| ||
��NOx��̼�⻯���������������·����⻯ѧ��Ӧ����������Ⱦ�������Ϊ���⻯ѧ����������ѧ�߶�ij���У����й⻯ѧ�����ı仯������вⶨ��ʵ������ͼ2����ͼ���֪��������Ⱦ����
ȩ��O3
ȩ��O3
�ȣ�ȩ��O3�ķ�ֵ������14��00���ҵ���Ҫԭ������ʱ�չ�ǿ�ң��⻯ѧ��Ӧ�������
��ʱ�չ�ǿ�ң��⻯ѧ��Ӧ�������
���۲ⶨ����β����Nox�ķ���֮һ����3%��H2O2��Һ����β���е�NOx����HNO3������NaOH����Һ�ζ�HNO3����Ҫ�ⶨ��״����β����NOx����������������������
ACD
ACD
��ѡ����ţ���A������β��������� B�����ֵ�������������
C��NaOH����Һ�����ʵ���Ũ�� D��������NaOH����Һ�����
E���ӵζ���ʼ��ָʾ����ɫ�����ʱ��
��3�����Ṥҵβ���е�NOx���ô�����Һ���գ��йصĻ�ѧ��ӦΪ��
2NO2+Na2O=NaNO3+CO2
NO+NO2+Na2CO3=2NaNO3+CO2
����0.5L 2mol/L�Ĵ�����Һǡ������һ�������Ṥҵβ���е�NOx������Һ��������44g����C02ȫ���ų�������NO2��NO�������Ϊ
7��1
7��1
����������1���ܷ�Ӧ����ʽ��д���ǽ��ַ�Ӧ����ʽ�ķ�Ӧ��ͷ�Ӧ�����һ�飬����������������һ�飬��Ӧ��������������ͬ�����ʷ���Ҫ�����ˣ���Ӧǰ��������ѧ���ʲ�����Ǵ�����
��2���ٸ���ͼ֪������Ӧ2NO+O2=2NO2��2NO2+4C0
4CO2+N2���ɵ��ܷ�Ӧ��
�ڶ�����Ⱦ����ָ���뻷���е�һ����Ⱦ������������ѧ���ػ�����������·����仯�����뻷���е��������ʷ�����Ӧ���γɵ���������ѧ��״��һ����Ⱦ�ﲻͬ������Ⱦ��ֳƼ̷�����Ⱦ��ݴ˼��ɽ��
�۸�����NaOH����Һ�ζ�HNO3���н��
��3�����ݶ�����̼�����������Һ�������ӵ����з�������һ�������Ͷ���������������Ӷ��ó������֮�ȣ�
��2���ٸ���ͼ֪������Ӧ2NO+O2=2NO2��2NO2+4C0
| ||
�ڶ�����Ⱦ����ָ���뻷���е�һ����Ⱦ������������ѧ���ػ�����������·����仯�����뻷���е��������ʷ�����Ӧ���γɵ���������ѧ��״��һ����Ⱦ�ﲻͬ������Ⱦ��ֳƼ̷�����Ⱦ��ݴ˼��ɽ��
�۸�����NaOH����Һ�ζ�HNO3���н��
��3�����ݶ�����̼�����������Һ�������ӵ����з�������һ�������Ͷ���������������Ӷ��ó������֮�ȣ�
����⣺��1����O3
O+O2����NO��NO2+O2����NO2+O��NO+O2���٢ڢ۷�Ӧ�з�Ӧ����O3��NO��NO2��O����������O��O2��NO2��NO������һ���ǣ�O3+NO+O+NO2�TO+3O2+NO����Ӧ�������������ͬ��Ҫ��ȥ����2O3=3O2���ɷ���ʽ��֪һ�������μ��˷�Ӧ�������������������˵�����Ǵ�����
�ʴ�Ϊ��2O3
3O2��NO��
��2���ٸ���ͼ֪������Ӧ����2NO+O2=2NO2 ����2NO2+4C0
4CO2+N2����+��ɵã�2NO+O2+4CO
4CO2+N2��
�ʴ�Ϊ��2NO+O2+4CO
4CO2+N2��
��һ����ȩͪ���������еĵ������̼�⻯�������չ������·����⻯ѧ��Ӧ���ɶ�����Ⱦ�����ȩ��O3�Ƕ�����Ⱦ���ͼ���֪����Ϊ���2�����ҹ��ߺ�ǿ�ң��䷴Ӧ�����ף�����ȩ��O3�ķ�ֵ������14��00���ң�
�ʴ�Ϊ��ȩ��O3����ʱ�չ�ǿ�ң��⻯ѧ��Ӧ������죻
����NOxת��ΪHNO3������NaOH����Һ�ζ�����NԪ���غ�֪n��NOx��=n��HNO3��������Ҫ��β����NOx�������������Ҫ�ⶨβ������������ζ����ĵ�NaOH����Һ������������ʵ���Ũ�ȣ�
�ʴ�Ϊ��ACD��
��3��2NO2+Na2CO3=NaNO2+NaNO3+CO2����m=48g��NO+NO2+Na2CO3=2NaNO3+CO2����m=32g����Ϸ�Ӧ����ʽ��֪����NOx�����ʵ���Ϊ2mol���γɵ�������Na��N��ͬʱ����1 mol CO2��0.5L 2mol/L�Ĵ�������ʵ�����1mol����NO�����ʵ�����x��NO2�����ʵ�����y����x+
=1mol��32x+24��y-x��=44����ã�x=
mol��Y=
mol��
��һ�������Ͷ������������ʵ���֮��=1��7��������ͬ�����£�����������һ�����������֮����7��1��
�ʴ�Ϊ��7��1��
| �� |
�ʴ�Ϊ��2O3
| ||
��2���ٸ���ͼ֪������Ӧ����2NO+O2=2NO2 ����2NO2+4C0
| ||
| ||
�ʴ�Ϊ��2NO+O2+4CO
| ||
��һ����ȩͪ���������еĵ������̼�⻯�������չ������·����⻯ѧ��Ӧ���ɶ�����Ⱦ�����ȩ��O3�Ƕ�����Ⱦ���ͼ���֪����Ϊ���2�����ҹ��ߺ�ǿ�ң��䷴Ӧ�����ף�����ȩ��O3�ķ�ֵ������14��00���ң�
�ʴ�Ϊ��ȩ��O3����ʱ�չ�ǿ�ң��⻯ѧ��Ӧ������죻
����NOxת��ΪHNO3������NaOH����Һ�ζ�����NԪ���غ�֪n��NOx��=n��HNO3��������Ҫ��β����NOx�������������Ҫ�ⶨβ������������ζ����ĵ�NaOH����Һ������������ʵ���Ũ�ȣ�
�ʴ�Ϊ��ACD��
��3��2NO2+Na2CO3=NaNO2+NaNO3+CO2����m=48g��NO+NO2+Na2CO3=2NaNO3+CO2����m=32g����Ϸ�Ӧ����ʽ��֪����NOx�����ʵ���Ϊ2mol���γɵ�������Na��N��ͬʱ����1 mol CO2��0.5L 2mol/L�Ĵ�������ʵ�����1mol����NO�����ʵ�����x��NO2�����ʵ�����y����x+
| y-x |
| 2 |
| 1 |
| 4 |
| 7 |
| 4 |
��һ�������Ͷ������������ʵ���֮��=1��7��������ͬ�����£�����������һ�����������֮����7��1��
�ʴ�Ϊ��7��1��
������������Ҫ�����˵����������֪ʶ�����ն��������Ļ�ѧ�����ǽ��Ĺؼ�����Ŀ�Ѷ��еȣ�

��ϰ��ϵ�д�
 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�
�����Ŀ
 ����������ᷢչ�����������ѳ�Ϊ������Ṳͬ��ע�Ľ��㣮
����������ᷢչ�����������ѳ�Ϊ������Ṳͬ��ע�Ľ��㣮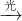 O+O2
O+O2 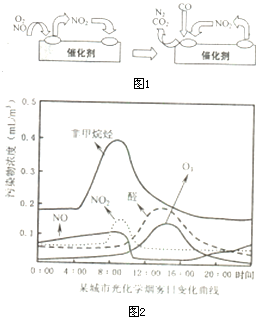
 O��O2 ��NO��O3��NO2��O2 ��NO2��O��NO��O2
O��O2 ��NO��O3��NO2��O2 ��NO2��O��NO��O2