��Ŀ����
��14�֣���ҵ����������β���ŷų���SO2��NOx�ȣ����γ���������Ҫ���ء������ɿ����еĻҳ������ᡢ���ᡢ�л�̼�⻯����������γɵ�������
��1�������е�SO2���̳��Ĵ����γ�����ķ�Ӧ����ʽ��____________________��
��2����֪2SO2 (g)+ O2 (g) 2SO3(g) ��H����196kJ/mol����߷�Ӧ��SO2��ת���ʣ��Ǽ���SO2�ŷŵ���Ч��ʩ��
2SO3(g) ��H����196kJ/mol����߷�Ӧ��SO2��ת���ʣ��Ǽ���SO2�ŷŵ���Ч��ʩ��
��T�¶�ʱ����2L�ݻ��̶�������ܱ������м���2.0 mol SO2��1.0 mol O2��5 min��Ӧ�ﵽƽ�⣬���������ת����Ϊ50%�����(O2)��____________��
���ڢٵ������£��жϸ÷�Ӧ�ﵽƽ��״̬�ı�־��_______(����ĸ)��
| A��SO2��O2��SO3���ߵ�Ũ��֮��Ϊ2��1��2 | B�������������ѹǿ���� |
| C�������ڻ��������ܶȱ��ֲ��� | D��SO3�����ʵ������ٱ仯 |
������Ӧ��ʼʱ���������м���1.5 mol SO2��0.8 mol O2����ƽ�����������ת���� ������ת���ʣ�����ڡ�С�ڻ���ڣ���
��3�������е�SO2������NaOH��Һ���գ������õ�Na2SO3��Һ���е�⣬��ѭ������NaOH��ͬʱ�õ�H2SO4����ԭ������ͼ��ʾ�����缫����Ϊʯī��
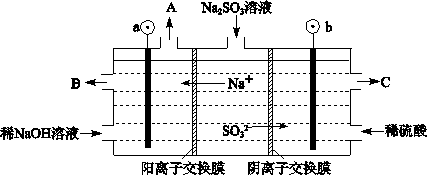
��ͼ��a��Ҫ���ӵ�Դ�ģ������������_______����C��������������_______��
��SO32���ŵ�ĵ缫��ӦʽΪ_____________________________________��
�۵�������������12.6gNa2SO3�����������仯������Ϊ_______g(����ù���������Һ��������ܱ�)��
��1��2SO2+2H2O+O2=2H2SO4 ��2����0.05mol/(L��min) �� BDE �۴���
��3���ٸ� ���� �� SO32���C2e����H2O��SO42��+2H+ ��4.4
���������������1�������е�SO2���̳��Ĵ����γ�����ķ�Ӧ����ʽ��2SO2+2H2O+O2=2H2SO4 ����2�����ڷ�Ӧ��ʼʱn(SO2) =2mol��5 min��Ӧ�ﵽƽ�⣬���������ת����Ϊ50%����Ӧ���ĵ�SO2�����ʵ���Ϊ1mol,��Ϊ�ڷ�ӦʱSO2�� O2�ǰ���2:1���ĵģ����Է�Ӧ�����������ʵ���Ϊ0.5mol,��˦�(O2)��0.5mol��2L��5 min=0.05mol/(L��min)����A���ڷ�Ӧ��SO2��O2��SO3�ǰ���2:1:2�Ĺ�ϵ�仯�ģ����Ǵﵽƽ��ʱ���ǵĹ�ϵ���ܷ�������������Ҳ���ܲ����ϡ���˲�����Ϊƽ��ı�־������B�����ڸ÷�Ӧ�ķ�Ӧǰ������������ȣ����ﵽƽ�⣬�������������ʵ������䣬ѹǿҲ���䡣��������������ѹǿ���������Ϊ�ж�ƽ��ı�־����ȷ��C�������������ݻ����䣬��Ӧ�ַ��������غ㶨�ɣ��������۷�Ӧ���е�ʲô�̶ȣ������ڻ��������ܶȶ����ֲ��䣬�ʲ�����Ϊ�ж�ƽ��ı�־������D������Ӧ�ﵽƽ�⣬�κ�һ�����ʵ����ʵ��������䣬Ũ��Ҳ���䣬���SO3�����ʵ������ٱ仯������Ϊ�ж�ƽ��ı�־����ȷ��E��ÿ����1��SO2�ͻ�ͬʱ����1��SO3�����ڵ�SO2�������ʺ�SO3������������ȣ���SO2�������ʺ�SO2������������ȣ���Ӧ�ﵽƽ�⡣��˿���Ϊ�ж�ƽ��ı�־ ����ȷ��������Ӧ��ʼʱ���������м���1.5 mol SO2��0.8 mol O2��n(SO2):n(O2)<2:1,���������˵����������ƽ�����������ת���ʴ���������ת���ʡ���3������ͼ������Na+��a�������ƶ�������ͬ�ֵ����ų⣬���ֵ���������ԭ��aҪ���ӵ�Դ�ĸ�����b���ӵ�Դ����������������SO32�����Ϸŵ��ΪSO42������˴�C�����������������ᡣ��SO32���ŵ�ĵ缫��ӦʽΪSO32���C2e����H2O��SO42��+2H+���۵�������������12.6gNa2SO3��n(Na2SO3)= 12.6g��126g/mol=0.1mol,�����ת��0.2mol,������������Ӧ��2H++2e-=H2��.�ų�����0.1mol,��������0.2g,ͬʱ���������0.2mold Na+,����Һ����4.6g,�ʸ����������������4.4g��
���㣺���黯ѧ��Ӧ���ʵļ��㡢ƽ��״̬���жϡ���ѧ����ʽ�����ӷ���ʽ����д���缫���жϼ��������� ��Һ�����ı仯�����֪ʶ��

����16�֣�����֪���з�Ӧ���Ȼ�ѧ����ʽΪ��
��1�� C(s) + O2(g) = CO2(g) ��H1 =" -393.5" kJ/mol
��2�� CH3COOH(l) + 2O2(g) = 2CO2(g) + 2H2O(l) ��H2 =" -870.3" kJ/mol
��3�� 2H2(g) + O2(g) = 2H2O(l) ��H3 =" -571.6" kJ/mol
�����:2C(s) + 2H2(g) + O2(g)= CH3COOH(l) ��H4 = ��
����ij�¶��£�����(t��BuNO)2���������CCl4�ܼ��о����Է�����Ӧ��
(t��BuNO)2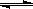 ��2(t��BuNO) �����¶��¸÷�Ӧ��CCl4�ܼ��е�ƽ�ⳣ��Ϊ1.4��
��2(t��BuNO) �����¶��¸÷�Ӧ��CCl4�ܼ��е�ƽ�ⳣ��Ϊ1.4��
��1����1L�������м���0.50mol��t-BuNO��2��10minʱ��Ӧ��ƽ�⣬��ʱ��t-BuNO��2��ƽ��ת����Ϊ60%�����跴Ӧ��������Һ���ʼ��Ϊ1L������Ӧ��ǰ10min�ڵ�ƽ������Ϊ�ͣ�t-BuNO��= ����ʽ����������Ӧ��ƽ�ⳣ��K =������ �� ��
��2���йط�Ӧ��(t��BuNO)2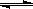 2(t��BuNO) ��������ȷ���ǣ� ��
2(t��BuNO) ��������ȷ���ǣ� ��
A��ѹǿԽ��Ӧ���ת����Խ�� B���¶����ߣ���ƽ��һ�������ƶ�
C���ܼ���ͬ��ƽ�ⳣ��Kֵ��ͬ
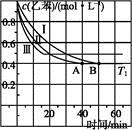
��3��ͨ����ɫ�����õ�40��ʱ(t��BuNO)2��(t��BuNO)Ũ����ʱ��ı仯��ϵ�ļ����������±���ʾ������ͬһͼ�л��(t��BuNO)2�ͣ�t��BuNO��Ũ����ʱ��ı仯���ߡ�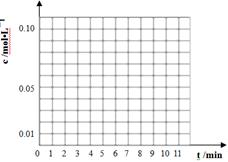
| ʱ�䣨min�� | 0 | 1 | 3 | 5 | 7 | 9 | 11 |
| c(t��BuNO)2 mol/L | 0.05 | 0.03 | 0.01 | 0.005 | 0.003 | 0.002 | 0.002 |
| c(t��BuNO) mol/L | 0 | 0.04 | 0.08 | 0.07 | 0.094 | 0.096 | 0.096 |
��ijʵ��С���H2O2�ķֽ���������̽�����±��Ǹ�ʵ��С���о�Ӱ��H2O2�ֽ����ʵ�����ʱ��¼��һ�����ݣ���������ͬ��״̬��ͬ��MnO2�ֱ����ʢ��15 ml 5%��H2O2��Һ�Ĵ��Թ��У����ô����ǵ�ľ�����ԣ�������£�
| MnO2 | �����Թ���� | �۲��� | ��Ӧ��������ʱ�� |
| ��ĩ״ | ���� | ���ҷ�Ӧ�������ǵ�ľ����ȼ | 3.5min |
| ��״ | �� | ��Ӧ���������Ǻ�����ľ��δ��ȼ | 30min |
��1��д�����Թ��з�����Ӧ�Ļ�ѧ����ʽ�� ���÷�Ӧ�� ��Ӧ������Ȼ����ȣ���
��2��ʵ���������������Ĵ�Ч���� �йء�
��ij���淴Ӧ��ij���Ϊ5L���ܱ������н��У� �ڴ�0��3���Ӹ����ʵ����ı仯�����ͼ��ʾ��A,B,C��Ϊ���壩��
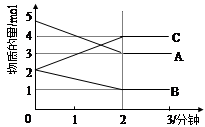
��3���÷�Ӧ�ĵĻ�ѧ����ʽΪ ��
��4����Ӧ��ʼ��2����ʱ��B��ƽ����Ӧ����Ϊ ��
��5����˵���÷�Ӧ�Ѵﵽƽ��״̬���� ��
a��v(A)= 2v(B) b��������ѹǿ���ֲ���
c��v��(A)= v��(C) d�������ڻ��������ܶȱ��ֲ���
��6����ͼ���A��ƽ��ʱ��ת����Ϊ ��
 CH3OCH3(g) + H2O(g)����֪һ�������£��÷�Ӧ��CO��ƽ��ת�������¶ȡ�Ͷ�ϱ�[n(H2) / n(CO)]�ı仯����������ͼ��
CH3OCH3(g) + H2O(g)����֪һ�������£��÷�Ӧ��CO��ƽ��ת�������¶ȡ�Ͷ�ϱ�[n(H2) / n(CO)]�ı仯����������ͼ��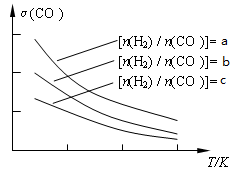
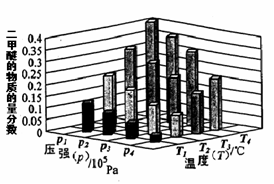




 xC��g��+2D��g��������5min��Ӧ�ﵽƽ��״̬�������ڵ�ѹǿ��С����֪D��ƽ����Ӧ����Ϊ0.2mol/��L?min��,����д���пհף�
xC��g��+2D��g��������5min��Ӧ�ﵽƽ��״̬�������ڵ�ѹǿ��С����֪D��ƽ����Ӧ����Ϊ0.2mol/��L?min��,����д���пհף� xC��g��+2D��g��������5min��Ӧ�ﵽƽ��״̬�������ڵ�ѹǿ��С����֪D��ƽ����Ӧ����Ϊ0.2mol/��L?min��,����д���пհף�
xC��g��+2D��g��������5min��Ӧ�ﵽƽ��״̬�������ڵ�ѹǿ��С����֪D��ƽ����Ӧ����Ϊ0.2mol/��L?min��,����д���пհף�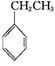 (g)+CO2(g)
(g)+CO2(g)
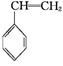 (g)+CO(g)+H2O(g)��H="-166" kJ��mol-1
(g)+CO(g)+H2O(g)��H="-166" kJ��mol-1