��Ŀ����
(9��)ijУһ��ѧʵ��С��ͨ��ʵ�������ȷ��һ��ɫ��ĩΪFe2O3��Cu2O�Ļ���
��֪:��Cu2O����ϡ��������Cu��CuSO4,��Cu2O�ڿ����м�������CuO��
(1)ʵ��С�����ü��ȷ��ⶨCu2O������������ȡa g�����ĩ�ڿ����г�ּ��ȣ����������ٱ仯ʱ����������Ϊb g(b>a)����������Cu2O����������Ϊ__________��
(2)ʵ��С�������øú�ɫ��ĩ��ȡ�ϴ����ĵ���(CuSO4��5H2O)�����������ϵ�֪������Һ��ͨ��������Һ������Զ�ʹCu2����Fe2����Fe3���ֱ����ɳ�����pH���£�
|
|
�������↑ʼ����ʱ��pH |
�������������ȫʱ��pH |
��ѡ���Լ� |
|
Fe3+ |
1.9 |
3.7 |
A.��ˮ�� B.H2O2�� C.NaOH�� D.��ˮ�� E.CuO�� F.Cu2(OH)2CO3 |
|
Fe2+ |
7.5 |
11 |
|
|
Cu2+ |
6.0 |
10 |
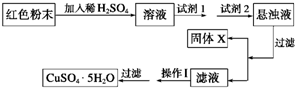
ʵ��С���������ͼʵ�鷽����
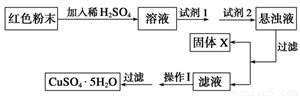
�Իش�
���Լ�1����Ϊ________________���Լ�2����Ϊ________________��
�ڹ���X�Ļ�ѧʽΪ____________��
�۲�����Ϊ____________________��
(1)��100%��3�֣�
(2)��B;��1�֣� EF����2�֣� ��Fe(OH)3��1�֣� ������Ũ������ȴ�ᾧ��2�֣�
����������1��������ͭ��������ͭ�ķ���ʽΪ2Cu2O��O2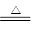 4CuO�����Ը��ݲ�������֪��������ͭ��������
4CuO�����Ը��ݲ�������֪��������ͭ��������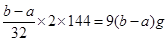 �����Ի������Cu2O����������Ϊ��100%��
�����Ի������Cu2O����������Ϊ��100%��
��2���ٻ������Һϡ�����У���������ͭ��ͭ��������������Ӧ�ü���˫��ˮ��������ͭ�����Լ�1��B���Լ�2��ԭ��������ҺpHֵ�ģ����ڲ����������ʣ����Է�Ӧ��EF��
��ͨ������pH������������������������X��Fe(OH)3��
��Ҫ�õ�����ͭ���壬Ӧ��ͨ������Ũ������ȴ�ᾧ�������˵õ���

 ��ѧʵ����ϵ�д�
��ѧʵ����ϵ�д�(9��)ijУһ��ѧʵ��С��ͨ��ʵ�������ȷ��һ��ɫ��ĩΪFe2O3��Cu2O�Ļ���
��֪:��Cu2O����ϡ��������Cu��CuSO4,��Cu2O�ڿ����м�������CuO��
(1)ʵ��С�����ü��ȷ��ⶨCu2O������������ȡa g�����ĩ�ڿ����г�ּ��ȣ����������ٱ仯ʱ����������Ϊb g(b>a)����������Cu2O����������Ϊ__________��
(2)ʵ��С�������øú�ɫ��ĩ��ȡ�ϴ����ĵ���(CuSO4��5H2O)�����������ϵ�֪������Һ��ͨ��������Һ������Զ�ʹCu2����Fe2����Fe3���ֱ����ɳ�����pH���£�
| | �������↑ʼ����ʱ��pH | �������������ȫʱ��pH | ��ѡ���Լ� |
| Fe3+ | 1.9 | 3.7 | A.��ˮ�� B.H2O2�� C.NaOH�� D.��ˮ�� E.CuO�� F.Cu2(OH)2CO3 |
| Fe2+ | 7.5 | 11 | |
| Cu2+ | 6.0 | 10 |
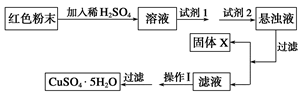
�Իش�
���Լ�1����Ϊ________________���Լ�2����Ϊ________________��
�ڹ���X�Ļ�ѧʽΪ____________��
�۲�����Ϊ____________________��

 ijУһ��ѧʵ��С��ͨ��ʵ�������ȷ��һ��ɫ��ĩΪFe2O3��Cu2O�Ļ���
ijУһ��ѧʵ��С��ͨ��ʵ�������ȷ��һ��ɫ��ĩΪFe2O3��Cu2O�Ļ���