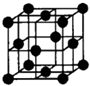
��Ŀ����
��9�֣���֪A��B��C��D��E�������ڱ���ǰ�����ڵ�Ԫ�أ����ǵĺ˵����A��B��C��D��E������A��B��C��ͬһ���ڵķǽ���Ԫ�ء�������DC�ľ���Ϊ���Ӿ��壬D�Ķ�����������C�������Ӿ�����ͬ�ĵ��Ӳ�ṹ��AC2Ϊ�Ǽ��Է��ӡ�B��C���⻯��ķе������ͬ����������Ԫ���⻯��ķе�ߡ�E��ԭ������Ϊ24��ECl3����B��C���⻯���γ�����λ��������������������ʵ���֮��Ϊ2��1������������λ����硣���������������ش��������⣺������ʱ��A��B��C��D��E������Ӧ��Ԫ�ط��ű�ʾ��
��1��A��B��C�ĵ�һ��������С�����˳��Ϊ ��
��2��B���⻯��ķ��ӿռ乹���� ��������ԭ�Ӳ�ȡ �ӻ���
��3��д��������AC2�ĵ���ʽ ��һ����B��C��ɵĻ�������AC2��Ϊ�ȵ����壬�仯ѧʽΪ ��
��4��E�ĺ�������Ų�ʽ�� ��ECl3�γɵ������Ļ�ѧʽΪ ��
��5��B������������Ӧ��ˮ�����ϡ��Һ��D�ĵ��ʷ�Ӧʱ��B����ԭ����ͼۣ��÷�Ӧ�Ļ�ѧ����ʽ�� ��
��1�� C<O<N (1��)
��2�� ���� (1��)�� sp3(1��)
��3��  (1��) �� N2O (1��)
(1��) �� N2O (1��)
��4�� 1s22s22p63s23p63d54s1����[Ar] 3d54s1�� (1��) �� [Cr(NH3)4(H2O)2]Cl3 (1��)
��5�� 4Mg+10HNO3=4Mg��NO3��2+NH4NO3+5H2O (2��) ��
��������
������������ݻ�����DC�ľ���Ϊ���Ӿ��壬D�Ķ�����������C�������Ӿ�����ͬ�ĵ��Ӳ�ṹ����֪D��Mg��C��O��AC2Ϊ�Ǽ��Է��ӣ���֪AΪC��BΪN��E��ԭ������Ϊ24����EΪCr��
��1������ͬ���ڵ�һ������������O��������C����O N��һ��������������
��2��NH3��SP3�ӻ������ӿռ乹���������͡�������ԭ�Ӳ�ȡsp3�ӻ���
��3��CO2�ĵ���ʽ�� ����N��O��ɵĻ�������CO2��Ϊ�ȵ����壬�仯ѧʽΪN2O��
����N��O��ɵĻ�������CO2��Ϊ�ȵ����壬�仯ѧʽΪN2O��
��4��Cr�ĺ�������Ų�ʽ��1s22s22p63s23p63d54s1����[Ar] 3d54s1��������Cr�Ŀչ������CrCl3�γɵ������Ļ�ѧʽΪ[Cr(NH3)4(H2O)2]Cl3��
��5��ϡ������Mg��Ӧʱ��N����ԭ����ͼ���-3�ۣ����ɵ���NH4+����÷�Ӧ�Ļ�ѧ����ʽ��4Mg+10HNO3=4Mg��NO3��2+NH4NO3+5H2O��
���㣺����Ԫ�ص��ƶϡ����ʽṹ�����ʵĹ�ϵ�������ܡ��ӻ�����ӹ��͡���ѧ����ʽ����д�ȡ�
��������Ԫ�����ڱ�ǰ20��Ԫ��Ҫ�������գ������ۺ��Խ�ǿ��

| A��Fe��Cu��Al��Ag | B��Al��Cu��Fe��Ag | C��Cu��Ag��Al��Fe | D��Ag��Al��Cu��Fe |

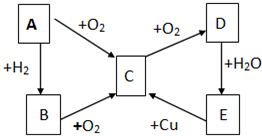 ��֪A��B��C��D��E������������ͼ��ʾ��ת����ϵ�����ַ�Ӧ�P��Ӧ����δ�г���������ʱ��Ҫ�������������裩�������������о�����AԪ�أ�
��֪A��B��C��D��E������������ͼ��ʾ��ת����ϵ�����ַ�Ӧ�P��Ӧ����δ�г���������ʱ��Ҫ�������������裩�������������о�����AԪ�أ� ����ͨ������£���A��BΪ���ӣ�C��EΪ�����ӣ�DΪ�����ӣ����Ƕ�����10�����ӣ�B����A�����õ����ʿɵ����C��D��A��B��E��������Ӧ��ɵ�C��һ�ְ�ɫ��������ش�
����ͨ������£���A��BΪ���ӣ�C��EΪ�����ӣ�DΪ�����ӣ����Ƕ�����10�����ӣ�B����A�����õ����ʿɵ����C��D��A��B��E��������Ӧ��ɵ�C��һ�ְ�ɫ��������ش�