��Ŀ����
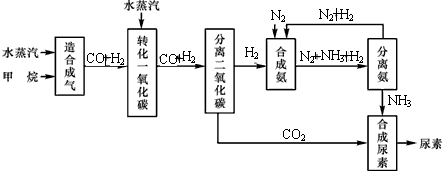
�Ĵ��зḻ����Ȼ����Դ������Ȼ��Ϊԭ�Ϻϳ����ص���Ҫ��������ͼ��ʾ(ͼ��ijЩת�����輰������δ�г�)��

����д���пհף�
(1)��֪0.5 mol������0.5 molˮ������t �桢p kPaʱ����ȫ��Ӧ����һ����̼���������ϳ�������������a kJ�������÷�Ӧ���Ȼ�ѧ����ʽ�ǣ� ��
(2)�ںϳɰ���ʵ�����������У�����ȡ�Ĵ�ʩ֮һ�ǣ������ɵİ��ӻ�������м�ʱ����������������������ĵ���������ѭ�����ã�ͬʱ���䵪���������������û�ѧ��Ӧ���ʺͻ�ѧƽ��Ĺ۵�˵����ȡ�ô�ʩ�����ɣ� ��
(3)������ϳɰ�����ת����Ϊ75%ʱ����5.60��107 L����Ϊԭ���ܹ��ϳ� L
������������������ڱ�״���²ⶨ��
(4)��֪���صĽṹ��ʽΪ![]() ����д�����ֺ���̼��˫�������ص�ͬ���칹��Ľṹ��ʽ��
����д�����ֺ���̼��˫�������ص�ͬ���칹��Ľṹ��ʽ��
�� ;�� ��
(1)CH4(g)+H2O(g)![]() CO(g)+3H2 (g);��H=2a kJ/mol
CO(g)+3H2 (g);��H=2a kJ/mol
(2)��������������Ũ������������Ӧ����;��С������Ũ�ȣ���������������Ũ�Ⱦ�������ƽ��������Ӧ�����ƶ�
(3)1.12��103
��4����![]() ��NH4N=C=O
��NH4N=C=O

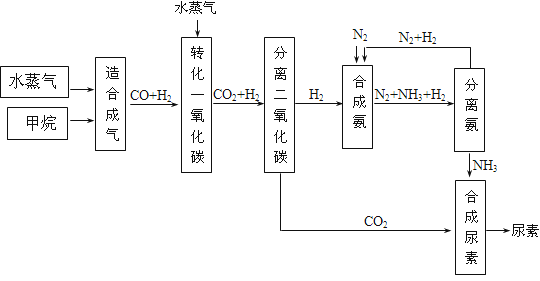
 ��������һ���þ�ϵ�д�
��������һ���þ�ϵ�д� Сѧ��10����Ӧ����ϵ�д�
Сѧ��10����Ӧ����ϵ�д�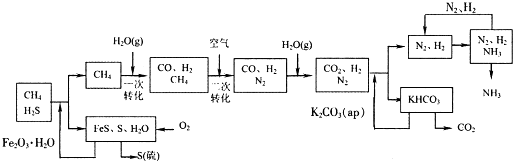
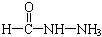

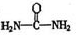 ����д�����ֺ���̼��˫�������ص�ͬ���칹��Ľṹ��ʽ����
����
��
����д�����ֺ���̼��˫�������ص�ͬ���칹��Ľṹ��ʽ����
����
��
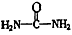 ����д�����ֺ���̼��˫�������ص�ͬ���칹��Ľṹ��ʽ����__________________ ����_______________________ ��
����д�����ֺ���̼��˫�������ص�ͬ���칹��Ľṹ��ʽ����__________________ ����_______________________ ��