��Ŀ����
������㷺Ӧ������ҩ�ͻ�����ҵ��ijͬѧ�����üױ���������Ӧ�Ʊ������ᡣ��Ӧԭ����
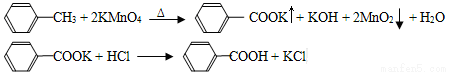
��֪�ױ����۵�Ϊ-95��C���е�Ϊ110.6��C���ӷ����ܶ�Ϊ0.866g/cm3����������۵�Ϊ122.4��C,��25��C��95��C���ܽ�ȷֱ�Ϊ0.3g��6.9g��
���Ʊ���Ʒ����30.0mL�ױ���25.0mLlmol/L���������Һ��100��C�·�Ӧ30min��װ����ͼ��ʾ��

��1��ͼ�������ܵĽ�ˮ��Ϊ________(�a����b������֧�ܵ�������___________��
��2���ڱ�ʵ���У�������ƿ����ʵ��ݻ�___________(����ĸ����
A��50mL | B��I00mL | C��200mL | D��250mL |
������þƾ���ֱ�Ӽ��ȣ��÷�ˮԡ���ȵ��ŵ���___________________��
�������Ʒ����ͬѧ����������̷���ֲ�Ʒ������ͻ��ռױ�
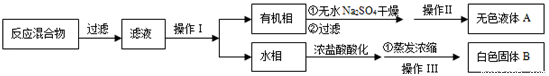
��3��������������� ��������־�IJ��ᆳ�������һ���ᴿ����ɫҺ��ױ����������������� ��
��4���ⶨ��ɫ������۵㣬��������115�濪ʼ�ۻ����ﵽ130��ʱ�����������ۣ���ͬѧ�Ʋ��ɫ�����DZ�������KCl�Ļ�����������·��������ᴿ�ͼ��飬ʵ���������Ʋ���ȷ������ɱ������ݡ�
��� | ʵ�鷽�� | ʵ������ | ���� |
�� | ����ɫ�������ˮ�У������ܽ⣬______�� | �õ���ɫ�������ɫ��Һ |
|
�� | ȡ������Һ���Թ��У�________�� | ���ɰ�ɫ���� | ��Һ��Cl- |
�� | �����ɫ���壬________�� | _____________________ | ��ɫ�����DZ����� |
���Ȳⶨ����ȡ1.220g��Ʒ�����100ml�״���Һ��ȡ25.00ml��Һ����0.1000mol/L KOH����Һ�ζ����ظ��ζ��ĴΣ�ÿ�����ĵ�������±���ʾ��
| ��һ�� | �ڶ��� | ������ | ���� |
�����mL�� | 24.00 | 24.10 | 22.40 | 23.90 |
��5���ζ������У������װ��KOH����Һ�ĵζ��ܶ���ʱ���ζ�ǰ���ӣ��ζ�������ⶨ���______________���ƫ����ƫС�����䡱����������Ʒ�б����ᴿ��Ϊ______________��
��ҵȼ��ú��ʯ�͵Ȼ�ʯȼ���ͷų�������������(NOx)��CO2��SO2�����壬������Ⱦ�������Է���������������̼����������ʵ����ɫ�������������á�
I.��������֪��H2����ֵΪ142.9 kJ/g(��ֵ�DZ�ʾ��λ������ȼ����ȫȼ��ʱ���ų�������)��
N2(g) + 2O2(g)= 2NO2(g) ��H= +133 kJ/mol
H2O(g) =H2O(l) ��H=- 44 kJ/mol
���������£�H2��ԭNO2����ˮ���������������ʵ��Ȼ�ѧ����ʽΪ_____________��
��.��̼����2L�ܱ������м���2 mol CO2��6 mol H2�����ʵ��Ĵ��������£�������Ӧ CO2(g) + 3H2(g)  CH3OH(l) + H2O(l)��
CH3OH(l) + H2O(l)��
��1���ٸ÷�Ӧ�Է����е�������___________(����¡������¡��������¶ȡ�����
������������˵���˷�Ӧ�ﵽƽ��״̬����___________��
A����������ƽ��ʽ�����ֲ���
B��CO2��H2������������ֲ���
C��CO2��H2��ת�������
D�����������ܶȱ��ֲ���
e.1 mol CO2���ɵ�ͬʱ��3 mol H��H������
��CO2��Ũ����ʱ��(0��t2)�ı仯��ͼ��ʾ����t2ʱ�������ݻ���Сһ����t3ʱ�ﵽƽ�⣬t4ʱ�����¶ȣ�t5ʱ�ﵽƽ�⣬�뻭��t2��t6��Ũ����ʱ��ı仯��

��2���ı��¶ȣ�ʹ��ӦCO2(g)+3H2(g) CH3OH(g) + H2O(g) ��H<0�е��������ʶ�Ϊ��̬����ʼ�¶������ͬ(T1�桢2L.�ܱ�����)����Ӧ�����в������ݼ��±���
CH3OH(g) + H2O(g) ��H<0�е��������ʶ�Ϊ��̬����ʼ�¶������ͬ(T1�桢2L.�ܱ�����)����Ӧ�����в������ݼ��±���
��Ӧʱ�� | CO2 (mol) | H2(mol) | CH3OH(mol) | H2O(mol) | |
��ӦI ���º��� | 0 min | 2 | 6 | 0 | 0 |
10 min | 4.5 | ||||
20 min | 1 | ||||
30 min | 1 | ||||
��ӦII���Ⱥ��� | 0 min | 0 | 0 | 2 | 2 |
�ٴﵽƽ��ʱ����ӦI��II�Աȣ�ƽ�ⳣ��K( I ) __________K(II)(�>����<����=������ͬ����ƽ��ʱCH3OH��Ũ��c(I)___ ________c(II)��
�ڶԷ�ӦI��ǰ10 min�ڵ�ƽ����Ӧ����v(CH3OH) =____________�����������������£���30 minʱֻ�ı��¶�ΪT2�棬��ʱH2�����ʵ���Ϊ3.2 mol����T1_________T2(���>����<����=��)����30 minʱֻ���������ٳ���1 mol CO2(g)��1mol H2O(g)����ƽ�� ______________�ƶ�(�������������)��


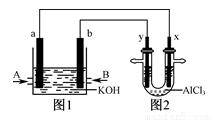
 Cl2����H2����2OH��
Cl2����H2����2OH��
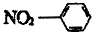
 �ɱ�ʾ������ӻ����Ȼ�̼����
�ɱ�ʾ������ӻ����Ȼ�̼���� 2NO ��g����O2 ��g�������ֲ�ͬ�����½��У�����ʵ�����800�棬ʵ�����850�棬NO��O2����ʼŨ�ȶ�Ϊ0��NO2��Ũ�� ��mol��L��1����ʱ�� ��min���ı仯��ͼ��ʾ����ش��������⣺
2NO ��g����O2 ��g�������ֲ�ͬ�����½��У�����ʵ�����800�棬ʵ�����850�棬NO��O2����ʼŨ�ȶ�Ϊ0��NO2��Ũ�� ��mol��L��1����ʱ�� ��min���ı仯��ͼ��ʾ����ش��������⣺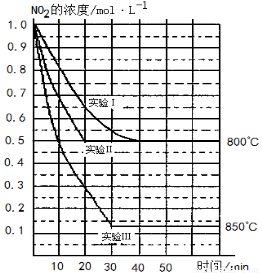
 ����Ⱦ���������������NH3��һ����������֮��Ӧ������ת��Ϊ���IJ������ѭ�������ʣ��磺8NH3+6NO2
����Ⱦ���������������NH3��һ����������֮��Ӧ������ת��Ϊ���IJ������ѭ�������ʣ��磺8NH3+6NO2 7N2+12H2O�������˷�Ӧ��Ƴ�ԭ��أ�����K2CO3Ϊ����ʣ���������Ӧ����ʽΪ�� ��
7N2+12H2O�������˷�Ӧ��Ƴ�ԭ��أ�����K2CO3Ϊ����ʣ���������Ӧ����ʽΪ�� �� ����ѧ�ͻ�ѧ���ܡ����й���̼���ܵ�˵������ȷ����
����ѧ�ͻ�ѧ���ܡ����й���̼���ܵ�˵������ȷ����
 ��������ͬ���칹���У��˴Ź���������ʾΪ2������________��д�ṹ��ʽ����ֻ������������ͬ���칹����__________�֡�
��������ͬ���칹���У��˴Ź���������ʾΪ2������________��д�ṹ��ʽ����ֻ������������ͬ���칹����__________�֡�