��Ŀ����
����������й�������Һ���ʱ�ı仯�����
(1)��ʯī���缫������ͼװ�õ��AlCl3��Һ���������������ݣ��������г������ɡ�������⣬��������������Һ�л��ɹ۲쵽�������� �����ʹ���������ӷ���ʽ�� ��
(2)����ʯī���缫���NaCl��Al2(SO4)3�Ļ����Һ�������Һ�ж��ߵ����ʵ���Ũ�ȷֱ�Ϊ3 mol��L-1��0.5 mol��L-1�������б�ʾ�����̵�������ȷ���� ��
(1)��ɫ�������ܽ�����ʧ Al(OH)3+OH-=AlO2-+2H2O
(2)A��D
����

��ѧ�����ͨѶ����ͨ���ճ����������Ź㷺��Ӧ�á�
��1��Ŀǰ���õ����ӣ�Ni��Cd����أ������ܷ�Ӧ���Ա�ʾΪ��
Cd��2NiO��OH����2H2O 2Ni��OH��2��Cd��OH��2
2Ni��OH��2��Cd��OH��2
��֪Ni��OH��2��Cd��OH��2��������ˮ���������ᣬ����˵����ȷ����________������ĸ��ţ���
�����Ϸ�Ӧ�ǿ��淴Ӧ�������Ϸ�Ӧ���ǿ��淴Ӧ���۳��ʱ��ѧ��ת��Ϊ���ܡ��ܷŵ�ʱ��ѧ��ת��Ϊ����
| A���٢� | B���ڢ� |
| C���٢� | D���ڢ� |
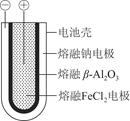
��3����һ�ֳ��õĵ��﮵�أ����һ�ּ����Ԫ�أ������ԭ������Ϊ7�����������ı���������λ�����缫��������ת���ĵ��ܣ��ر����㷺Ӧ��������������һ��ʹ��ʱ��ɳ���ʮ�꣬���ĸ����ɽ�����Ƴɣ�����ܷ�Ӧ�ɱ�ʾΪLi��MnO2=LiMnO2��
�Իش�﮵�ر������ر���ԭ����___________________________��
﮵���еĵ������Һ���÷�ˮ��Һ���ƣ�Ϊʲô���ֵ�ز���ʹ�õ���ʵ�ˮ��Һ�����û�ѧ����ʽ��ʾ��ԭ��___________________________________________________��
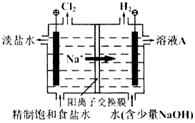
��1���������ʵ�����KI��CuCl2����ˮ���ö��Ե缫��⣬�õ�ⷴӦ�ɷ�Ϊ________���Σ�����һ���������������������������ӣ���
| �� | �൱�ڵ��ʲô��Һ | ���ӷ���ʽ |
| �� | | |
| �� | | |
| �� | | |
| �� | | |
| �� | | |
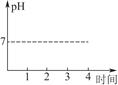
��2��������������ҺpH��ʱ��仯�����ߣ��ٶ����ɵ�Cl2ȫ���ݳ�����



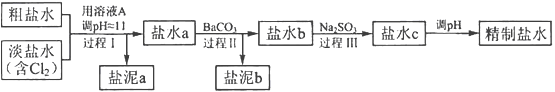
 Fe��2NaCl���õ�طŵ�ʱ��������ӦʽΪ________________________________________________________________________��
Fe��2NaCl���õ�طŵ�ʱ��������ӦʽΪ________________________________________________________________________��