��Ŀ����
��NA��ʾ�����ӵ�������ֵ������˵������ȷ��
��17.6 g�����������Ĺ��ۼ���Ϊ4NA��
�ڳ����£�21 g C��H����C��H���Ļ�����к��е�̼ԭ����Ϊ1.5NA
�۵�ⱥ��ʳ��ˮ������������2.24 L H2ʱ��ת�Ƶĵ�����Ϊ0.2NA
��2 g D������O�к��е�������������������������ΪNA
�ݳ����£�1.0 L pH��13��NaOH��Һ�У���ˮ�����OH��������ĿΪ0.1NA
��100 mL0.1 mol��L������FeCl����Һ�����ˮ�п��Ƶ�Fe(OH)������0.01NA
A. �٢ڢ� B. �٢ۢ�
C. �ܢݢ� D. �٢�
��ϰ��ϵ�д�
 ��������һ���þ�ϵ�д�
��������һ���þ�ϵ�д� Сѧ��10����Ӧ����ϵ�д�
Сѧ��10����Ӧ����ϵ�д�
�����Ŀ



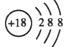 D. ��ϩ�Ľṹ��ʽ��CH2CH2
D. ��ϩ�Ľṹ��ʽ��CH2CH2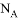 Ϊ����٤��������ֵ������������ȷ����
Ϊ����٤��������ֵ������������ȷ���� ��
��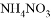 ��Һ�е�ԭ����Ϊ0.2
��Һ�е�ԭ����Ϊ0.2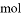
 ת�Ƶĵ�����Ϊ6
ת�Ƶĵ�����Ϊ6