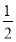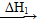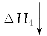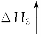��Ŀ����
��ҵ����������Ϊԭ����������������β�����˺���N2��O2�⣬������SO2������SO3��������Ϊ�˱���������ͬʱ������Ṥҵ���ۺϾ���Ч�棬Ӧ�����ܽ�β���е�SO2ת��Ϊ���õĸ���Ʒ���밴Ҫ��ش��������⣺
(1)��β��ͨ�백ˮ�У��ܷ��������Ӧ��д�����п��ܷ���������������ԭ��Ӧ�Ļ�ѧ����ʽ��__________________��__________________��
(2)��β���백ˮ��Ӧ���õ��ĸ�Ũ����Һ�У���һ ���������백ˮ��̼����泥���ʱ��Һ���¶Ȼ����н��ͣ����������塣��������Һ�¶Ƚ��͵�ԭ�������______________���������ľ����������ֽ��ҵ��Ҳ��������������ӰҺ����������֪�ýᾧˮ�������Է�������Ϊ134�����仯ѧʽΪ______________��
(1)2H2SO3��O2=2H2SO4��2(NH4)2SO3��O2=2(NH4)2SO4��
2NH4HSO3��O2=2NH4HSO4(��������)
(2)����Ӧ���ȡ���(NH4)2SO3��H2O
��������(1)����Ҫע����ĿҪ�����д������������ԭ��Ӧ�ķ���ʽ(����)��
(2)����Һ�¶Ƚ���һ���Ƿ�Ӧ�������£��������ľ���������ֽ����ӰҺ��˵�������к��л�ԭ�ԣ�4���������֪�������Ҫ�ɷ�Ϊ(NH4)2SO3��������Է�������Ϊ134�����Ƴ��仯ѧʽΪ(NH4)2SO3��H2O��