��Ŀ����
��1������ɫ�Լ���˫��ˮ����Ϊ��ҵ��Һ���������������ɿ�ҵ��Һ�е��軯���KCN������Ӧ�Ļ�ѧ����ʽΪ��KCN+H2O2+H2O��A+NH3��
�ش��������⣺
��������A�Ļ�ѧʽΪ____________��
���ڱ�״��������0.448L�������ɣ���ת�Ƶĵ�����Ϊ____________��
�۷�Ӧ�б�������Ԫ��Ϊ____________��
��2����MgCl2��AlCl3�Ļ����Һ�У���μ���NaOH��Һֱ �����������ⶨ�������NaOH�����ʵ��������ó��������ʵ����Ĺ�ϵ����ͼ��ʾ����
�����������ⶨ�������NaOH�����ʵ��������ó��������ʵ����Ĺ�ϵ����ͼ��ʾ����

��ԭ��Һ ��c��Mg2������c��Al3����Ϊ ��
��c��Mg2������c��Al3����Ϊ ��
��ͼ��C���ʾ������ mol NaOHʱ��
Al3���Ѿ�ת��Ϊ���� �� ����
Mg2���Ѿ�ת��Ϊ ��
��ͼ���߶�OA��AB�� ��
�ش��������⣺
��������A�Ļ�ѧʽΪ____________��
���ڱ�״��������0.448L�������ɣ���ת�Ƶĵ�����Ϊ____________��
�۷�Ӧ�б�������Ԫ��Ϊ____________��
��2����MgCl2��AlCl3�Ļ����Һ�У���μ���NaOH��Һֱ
 �����������ⶨ�������NaOH�����ʵ��������ó��������ʵ����Ĺ�ϵ����ͼ��ʾ����
�����������ⶨ�������NaOH�����ʵ��������ó��������ʵ����Ĺ�ϵ����ͼ��ʾ����
��ԭ��Һ
 ��c��Mg2������c��Al3����Ϊ ��
��c��Mg2������c��Al3����Ϊ ����ͼ��C���ʾ������ mol NaOHʱ��
Al3���Ѿ�ת��Ϊ���� �� ����
Mg2���Ѿ�ת��Ϊ ��
��ͼ���߶�OA��AB�� ��
��1����KHCO3 ��2.408��1022 ��KCN�е�C��̼��
��2����2��1
 �� 0��8 NaAlO2 Mg��OH��2
�� 0��8 NaAlO2 Mg��OH��2
��7��1
��2����2��1
 �� 0��8 NaAlO2 Mg��OH��2
�� 0��8 NaAlO2 Mg��OH��2��7��1
��

��ϰ��ϵ�д�
�����Ŀ
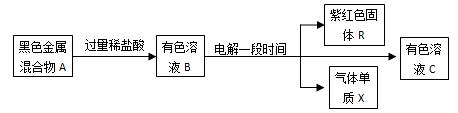



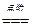 2HCl ��Na2CO3+2HCl==2NaCl+H2O+CO2��
2HCl ��Na2CO3+2HCl==2NaCl+H2O+CO2��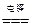 2H2��+O2�� ��CuO+2HNO3==Cu(NO3)2+H2O ��2HgO
2H2��+O2�� ��CuO+2HNO3==Cu(NO3)2+H2O ��2HgO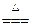 2Hg+O2��
2Hg+O2��