��Ŀ����
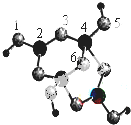
18����1��һ��������ij����H2�����ʵ���֮��1��2�ӳ�����CH3CH2CH��CH3��2��������Ľṹ��ʽΪ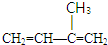 ��
��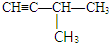 ��
����2��ij�л���X������ֻ��C��H��O����Ԫ�أ���Է�������С��110��������Ԫ�ص���������Ϊ14.8%����֪�����ʿ���FeCl3��Һ������ɫ��Ӧ����X�ķ���ʽΪC7H8O����1mol X��Ũ��ˮ��Ӧʱ������3mol Br2����X�Ľṹ��ʽΪ
 ������ʽΪC4H8�������˴Ź�������ͼ�����������շ壬�������Ϊ3��1����д�������������������ʵĽṹ��ʽ
������ʽΪC4H8�������˴Ź�������ͼ�����������շ壬�������Ϊ3��1����д�������������������ʵĽṹ��ʽ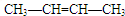 ��
��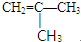
��֪��
 �����Ҫ�ϳ�
�����Ҫ�ϳ�  ���õ�ԭʼԭ�Ͽ�����AC��
���õ�ԭʼԭ�Ͽ�����AC��A��2-��-l��3-����ϩ��2-��Ȳ B��2��3-����-1��3-���ϩ����Ȳ
C��2��3-����-l��3-����ϩ�ͱ�Ȳ D��1��3-���ϩ��2-��Ȳ��
���� ��1��ij����H2�����ʵ���֮��1��2�ӳɣ�������к���̼̼����������̼̼˫�����ݴ�д���������ܵĽṹ��ʽ��
��2��������Ԫ�غ���ȷ�����л�����Է��������ٸ��ݸ��л���Ļ�ѧ�����ж��ж������ʽ���ٸ��ݡ�1mol X��Ũ��ˮ��Ӧʱ������3molBr2���ж�ȡ����λ�ã�Ȼ��д�����ܽṹ��ʽ�����ݡ��˴Ź�������ͼ�����������շ壬�������Ϊ3��1���жϺ��е�Ч��ԭ�����༰��Ŀ���ٽ�Ϸ���ʽC4H8д����������Ľṹ��ʽ��
��3���������з�Ӧԭ������Ӧ���� ���ж�Ȳ���ṹ��Ȼ���ж϶�ϩ�������ƣ��Ӷ��ó���ȷ���ۣ�
���ж�Ȳ���ṹ��Ȼ���ж϶�ϩ�������ƣ��Ӷ��ó���ȷ���ۣ�
��� �⣺��1��һ��������ij����H2�����ʵ���֮��1��2�ӳ�����CH3CH2CH��CH3��2���������2��˫����1������������ܵĽṹ��ʽΪ��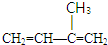 ��
��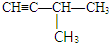 ��
��
�ʴ�Ϊ��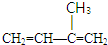 ��
��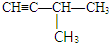 ��
��
��2������FeCl3��Һ������ɫ��Ӧ����˵�����з��ǻ�����X��Է�������С��110����Ԫ�ص���������Ϊ14.8%��������1��Oԭ��ʱ������Է�����Ϊ��$\frac{16}{14.8%}$��108������2��Oԭ��ʱ������Ϊ216��110������л��������ֻ��1���ǻ������ӵ�ʽ��Ϊ��94�����л���ʽ��Ϊ108��108-94=12���뱽�����14�������һ��CH2������л���ķ���ʽΪ��C7H8O��
��1 mol X��Ũ��ˮ��Ӧʱ������3 mol Br2��˵�����ǻ�����λû��ȡ������������ǻ��ļ�λ�����л���Ľṹ��ʽΪ�� ��
��
����ʽΪC4H8�������˴Ź�������ͼ�����������շ壬�������Ϊ3��1��˵�������д�������Hԭ�ӣ��Ҹ�����Ϊ3��1������ܵĽṹ��ʽΪ��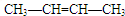 ��
��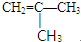 ��
��
�ʴ�Ϊ��C7H8O�� ��
��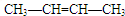 ��
��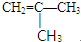 ��
��
��3������������Ϣ��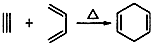 ����֪���ϳ�
����֪���ϳ� ��Ȳ�������ǣ�2-��Ȳ���Ȳ����Ϊ2-��Ȳ�����ϩ��Ϊ2-��-l��3-����ϩ����Ϊ��Ȳ�����ϩ��Ϊ2��3-����-l��3-����ϩ������A��C��ȷ��
��Ȳ�������ǣ�2-��Ȳ���Ȳ����Ϊ2-��Ȳ�����ϩ��Ϊ2-��-l��3-����ϩ����Ϊ��Ȳ�����ϩ��Ϊ2��3-����-l��3-����ϩ������A��C��ȷ��
�ʴ�Ϊ��AC��
���� ���⿼�����л������ʽ���ṹ��ʽ��ȷ������Ŀ�Ѷ��еȣ����������ϴ��漰��֪ʶ��϶࣬ע�������л������ʽ���ṹ��ʽ�ķ�������������������ѧ���ķ������������������Ӧ��������

| A�� | NH4NO3����ˮ���ȣ�˵��������ˮ�����Է����� | |
| B�� | ͬһ������̬ʱ��ֵ���Һ̬ʱ��֮������̬ʱ��ֵ��С | |
| C�� | ���ʷ���������ԭ��Ӧ����������ת�� | |
| D�� | �������оݺ����о���ϳɵĸ����оݣ����ʺ������еĹ��� |
��1��N��Al��Si��Zn����Ԫ���У���һ��Ԫ�صĵ������������£�
| ������ | I1 | I2 | I3 | I4 | �� |
| Im/kJ•mol-1 | 578 | 1 817 | 2 745 | 11 578 | �� |
��2���ࣨGe�����ܵ����ʻ�Ӧ����CD_��
A����һ�ֻ��õĽ���Ԫ�� B����縺�Դ�����
C���䵥�ʿ���Ϊ�뵼����� D��������Ȼ���ķе�������廯��ķе�
��3�����ڻ�����HOH2CCH2OH������������ȷ����ABD��
A�����Ӽ���γ���� B�������м��м��Լ����зǼ��Լ�
C����������7���Ҽ���1���м� D���÷�����ˮ�е��ܽ�ȴ���2-��ϩ
��4��NaF������۵㣾[
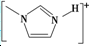 ]BF${\;}_{4}^{-}$���۵㣨���=������ԭ�������߾�Ϊ���ӻ�����ҵ������Ϊ1�����������Ӱ뾶�����Ӽ�����������۵�ϵͣ�
]BF${\;}_{4}^{-}$���۵㣨���=������ԭ�������߾�Ϊ���ӻ�����ҵ������Ϊ1�����������Ӱ뾶�����Ӽ�����������۵�ϵͣ� | A�� | 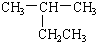 2-�һ����� 2-�һ����� | B�� | 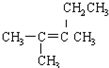 2-��-3-�һ���ϩ 2-��-3-�һ���ϩ | ||
| C�� | H5C2OOCCOOC2H5 �Ҷ����Ҷ��� | D�� | CH3CH2CH2CH2OH 1-���� |
| A�� | CH2=CH2��CH2=CH-CH=CH2 | B�� | CH��CH�� CH3-CH2-C��CH | ||
| C�� | CH3OH��CH3CH2OH | D�� |  �� ��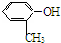 |
��
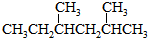 ��
��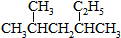 ��CH3CH2CH3
��CH3CH2CH3��HC��CCH3 ��
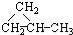 ��CH3CH=CHCH3��
��CH3CH=CHCH3��| A�� | �ٺ͢� | B�� | �ٺ͢� | C�� | �ٺ͢� | D�� | �ݺ͢� |
| A�� | ��CO2������ʯ+ϡ���� | B�� | ��SO2��Na2SO3���̣�+Ũ���� | ||
| C�� | ��H2��Fe���̣�+ϡ���� | D�� | ��Cl2��MnO2���̣�+ϡ���� |
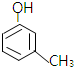
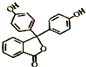 ����ṹ�Ͽ�����̪���ڷӻ����������
����ṹ�Ͽ�����̪���ڷӻ����������