��Ŀ����
��֪Ԫ�صĵ縺�Ժ�ԭ�Ӱ뾶�������ԡ��ǽ����Ե�һ����Ҳ��Ԫ�ص�һ�ֻ������ʡ��������13��Ԫ�ص縺�Ե���ֵ
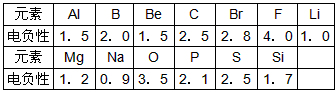
�Խ��Ԫ��������֪ʶ����������⣺
��1�����ƶ�AlBr3��NaF�еĻ�ѧ�����ͷֱ���_________��_________��
��2�������ϱ����������ݣ���������Ԫ�ص縺�Ե���ֵ��С��Ԫ�صĽ����Ի�ǽ�����ǿ��֮��Ĺ�ϵ______________________________�������ڶ�����Ԫ��(������������)�縺�Ե���ֵ��С��ԭ�Ӱ뾶֮��Ĺ�ϵ___________________��
��3������Ԥ��Br��IԪ�صĵ縺�ԵĴ�С��ϵBr____I��
��4��ij�л�����������к���S-N��������Ϊ�ù��õ��Ӷ�ƫ����______ԭ��(��Ԫ�ط���)��
��1�����ƶ�AlBr3��NaF�еĻ�ѧ�����ͷֱ���_________��_________��
��2�������ϱ����������ݣ���������Ԫ�ص縺�Ե���ֵ��С��Ԫ�صĽ����Ի�ǽ�����ǿ��֮��Ĺ�ϵ______________________________�������ڶ�����Ԫ��(������������)�縺�Ե���ֵ��С��ԭ�Ӱ뾶֮��Ĺ�ϵ___________________��
��3������Ԥ��Br��IԪ�صĵ縺�ԵĴ�С��ϵBr____I��
��4��ij�л�����������к���S-N��������Ϊ�ù��õ��Ӷ�ƫ����______ԭ��(��Ԫ�ط���)��
��1�����ۼ������Ӽ�
��2��Ԫ�صĵ縺����ֵԽ��Ԫ�صķǽ�����Խǿ(��Ԫ�صĵ縺����ֵԽС��Ԫ�صĽ�����Խǿ)��ԭ�Ӱ뾶ԽС���縺�Ե���ֵԽ��
��3��Br����I
��4��N
��2��Ԫ�صĵ縺����ֵԽ��Ԫ�صķǽ�����Խǿ(��Ԫ�صĵ縺����ֵԽС��Ԫ�صĽ�����Խǿ)��ԭ�Ӱ뾶ԽС���縺�Ե���ֵԽ��
��3��Br����I
��4��N

��ϰ��ϵ�д�
 �㽭��У��ʦ���ϵ�д�
�㽭��У��ʦ���ϵ�д�
�����Ŀ
 ��2011?����һģ�������ڳ�ȥ���ٹ�·��ѩ���ǡ������ࡱ��ѩ������NaCl��MgCl2�ȣ���ش�
��2011?����һģ�������ڳ�ȥ���ٹ�·��ѩ���ǡ������ࡱ��ѩ������NaCl��MgCl2�ȣ���ش� ��2011?���գ�W��X��Y��Z�����ֳ����Ķ�����Ԫ�أ���ԭ�Ӱ뾶��ԭ�������仯����ͼ��ʾ����֪W��һ�ֺ��ص�������Ϊ18��������Ϊ10��X��Neԭ�ӵĺ�����������1��Y�ĵ�����һ�ֳ����İ뵼����ϣ�Z�ĵ縺����ͬ��������Ԫ�������
��2011?���գ�W��X��Y��Z�����ֳ����Ķ�����Ԫ�أ���ԭ�Ӱ뾶��ԭ�������仯����ͼ��ʾ����֪W��һ�ֺ��ص�������Ϊ18��������Ϊ10��X��Neԭ�ӵĺ�����������1��Y�ĵ�����һ�ֳ����İ뵼����ϣ�Z�ĵ縺����ͬ��������Ԫ�������
 ��101 kPa�£���֪Y����̬��������������ȫȼ�պ�ָ���ԭ״̬��ƽ��ÿת��1mol ���ӷ���190.0kJ���÷�Ӧ���Ȼ�ѧ����ʽ��
��
��101 kPa�£���֪Y����̬��������������ȫȼ�պ�ָ���ԭ״̬��ƽ��ÿת��1mol ���ӷ���190.0kJ���÷�Ӧ���Ȼ�ѧ����ʽ��
�� A+ N2�� +3CO2��
A+ N2�� +3CO2��