��Ŀ����
��ǰ����������һ��ȫ�����ӵ����⣬������������峣��������������CO2��
��Ⱦ������NOx��SOx�ȡ��������Щ����������þͿ��Գ�Ϊ��Ҫ����Դ���Ƚ���˶Ի�������Ⱦ���ֽ���˲�����ԴΣ�����⡣
(1)������̼�ǵ�������ЧӦ��������ף�Ŀǰ���Ǵ���������̼�ķ���֮һ��ʹ����������Ӧ�ϳɼ״����״�������ȼ�ϵ�ص���Ҫȼ�ϡ�CO2��H2��Ӧ�Ʊ�CH3OH��H2O�Ļ�ѧ����ʽΪ
(2)�ڸ�����һ����̼�ɽ���������ԭΪ������
��֪��
��C(s)��O2(g)=CO2(g)��H1����393.5 kJ��mol��1
��CO2(g)��C(s)=2CO(g)��H2����172.5 kJ��mol��1
��S(s)��O2(g)=SO2(g)��H3����296.0 kJ��mol��1
��д��CO��SO2��Ӧ���Ȼ�ѧ����ʽ ��
(3)���᳧���ô���ԭ��������β����CH4�ڴ������¿��Խ�NO2��ԭΪN2��
��֪��CH4(g)��2O2(g)=CO2(g)��2H2O(g)��H����889.6 kJ��mol��1��
N2(g)��2O2(g)=2NO2(g)��H����67.7 kJ��mol��1��
��CH4��ԭNO2����ˮ�����͵������Ȼ�ѧ����ʽ�� ��
(1)CO2��3H2=CH3OH��H2O
(2)2CO(g)��SO2(g)=S(s)��2CO2(g) ��H����270 kJ��mol��1
(3)CH4(g)��2NO2(g)=N2(g)��2H2O(g)��CO2(g) ��H����957.3 kJ��mol��1
����

 �����ߴ���ϵ�д�
�����ߴ���ϵ�д���ͼ�Ǻ���P��s����Cl2��Ӧ����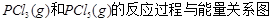 ��ͼ�е�
��ͼ�е� ��ʾ����1mol��������ݣ���������ͼ�ش��������⣺
��ʾ����1mol��������ݣ���������ͼ�ش��������⣺
��1��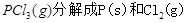 ���Ȼ�ѧ����ʽΪ ��
���Ȼ�ѧ����ʽΪ ��
��2��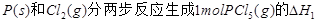 = KJ��mol-1
= KJ��mol-1
��3���о���������ѧ��Ӧ�������仯����H���뷴Ӧ���������ļ����йء����ܿ��Լ�����Ϊ�Ͽ�1mol��ѧ��ʱ�������յ���������1�����Dz��ֻ�ѧ���ļ������ݡ�
��1���ֻ�ѧ���ļ�������
| ��ѧ�� | P-P | P-O | O=O | P=O |
| ����/��kJ��mol-1�� | 198 | 360 | 498 | x |
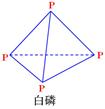
��֪1mol���ף��ṹ����ͼ��ʾ������ʽΪP4����ȫȼ������P4O10���ṹ����ͼ���ų�2982KJ����������У�x= ��
 ��P4O10��
��P4O10��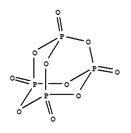
�Ҵ����ͺ�������35%,ʹȼ��ȼ�ո��ӳ��,ʹ�ó����Ҵ����ͣ�β���ŷŵ�CO��̼�⻯����ƽ������30%����,��Ч�Ľ��ͺͼ������к���β���ŷš���������ʹ���Ҵ����Ͳ����ܼ���NOx���ŷţ���NOx����Ч������Ϊ�����������Ҫ���⡣NOx��������У��γ����꣬��ɿ�����Ⱦ��NOx����һ�ֺ���ɫ���壬������ˮ�ķ���ʽ�� ��
��2����֪NO2��N2O4�Ľṹʽ�ֱ���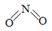 ��
��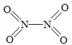 ��
��
| ���� | NO2 | N2O4 | |
| ��ѧ�� | N��O | N��N | N��O |
| ���ܣ�kJ/mol�� | 466 | 167 | 438 |
д��NO2ת��N2O4���Ȼ�ѧ����ʽ ��
��3���о���Ա������β��ϵͳ��װ�ô�ת����������Ч����NOx���ŷš�
�� д����CO��ԭNO����N2�Ļ�ѧ����ʽ ��
�� ��ʵ������ģ�´˷�Ӧ����һ�������µ��ܱ������У����NOת��ΪN2��ת�������¶ȱ仯�����n (NO)/n(CO)�����仯�������ͼ��
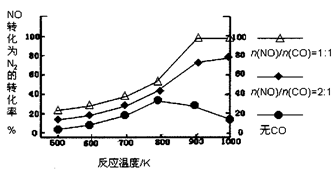
Ϊ�ﵽNOת��ΪN2�����ת���ʣ�Ӧ��ѡ�õ��¶Ⱥ�n(NO)/n(CO)�����ֱ�Ϊ �� ���÷�Ӧ��?H 0�����������������������
��4���� CxHy(��)����ԭNOxҲ����������������������Ⱦ�����ʡ�CH4��NO������Ӧ�Ļ�ѧ����ʽΪ ��
̼�͵��Ļ���������������������������ء�
��1����һ���¡������ܱ������з�����Ӧ�� Ni(s)+4CO(g) Ni(CO)4(g)��
Ni(CO)4(g)�� H<0�����ø÷�Ӧ���Խ�����ת��Ϊ���ȴ�99��9���ĸߴ������Ը÷�Ӧ��˵����ȷ���� (����ĸ���)��
H<0�����ø÷�Ӧ���Խ�����ת��Ϊ���ȴ�99��9���ĸߴ������Ը÷�Ӧ��˵����ȷ���� (����ĸ���)��
| A������Ni���������CO��ת���ʣ�Ni��ת���ʽ��� |
B����С�����ݻ���ƽ�����ƣ� H��С H��С |
| C����Ӧ�ﵽƽ�����CO�ٴδﵽƽ��ʱ��CO������������� |
| D����4v[Ni(CO)4]=v(CO)ʱ�������л�������ܶȲ���ʱ������˵����Ӧ�Ѵﻯѧƽ��״̬ |
��֪��C(s)+
 O2(g)=CO(g)
O2(g)=CO(g)  H=-Q1 kJ
H=-Q1 kJ mol-1
mol-1C(s)+ O2(g)=CO2(g)
 H=-Q2 kJ
H=-Q2 kJ mol-1
mol-1S(s)+O2(g)=SO2(g)
 H=-Q3 kJ
H=-Q3 kJ mol-1
mol-1��SO2(g)+2CO(g)=S(s)+2CO2(g)
 H= ��
H= ����3������������ɱ�һ����̼��ԭ���ɽ������ʺͶ�����̼��ͼ28��3�������ֽ��������Cr2O3��SnO2��PbO2��Cu2O)��һ����̼��ԭʱ
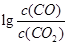 ���¶ȣ�t���Ĺ�ϵ����ͼ��
���¶ȣ�t���Ĺ�ϵ����ͼ��700oCʱ���������ѱ���ԭ�Ľ����������� (�ѧʽ)����һ����̼��ԭ�ý���������ʱ������Ӧ����ʽϵ��Ϊ��������ȣ��÷�Ӧ��ƽ�ⳣ��(K)��ֵ���� ��

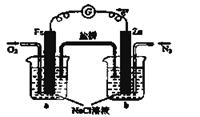
��4��NO2��O2������NaNO3������ȼ�ϵ�أ���ԭ������ͼ28��4����ʾ���õ����ʹ�ù�����ʯīI�缫������������Y����缫��ӦʽΪ ��
����ȼ�ϵ��ʹ��һ��ʱ����ռ���20mol Y������������Ҫ���ı�״�������������Ϊ L��
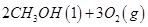 =
=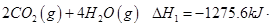 mol
mol
 =
=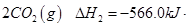 mol
mol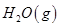 =
=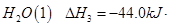 mol
mol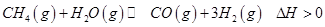
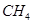 ��ƽ��ת�������¶ȡ�ѹǿ�Ĺ�ϵ��ͼa����
��ƽ��ת�������¶ȡ�ѹǿ�Ĺ�ϵ��ͼa����
 (�<������>����="����ͬ)��A��B��C���㴦��Ӧƽ�ⳣ��(
(�<������>����="����ͬ)��A��B��C���㴦��Ӧƽ�ⳣ��(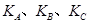 )�Ĵ�С��ϵΪ ��
)�Ĵ�С��ϵΪ ��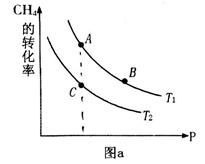
 ͨ���ݻ�Ϊ1L�Ķ����ܱ������з�����Ӧ����˵���÷�Ӧ�Ѿ��ﵽƽ��״̬���� (�����)��
ͨ���ݻ�Ϊ1L�Ķ����ܱ������з�����Ӧ����˵���÷�Ӧ�Ѿ��ﵽƽ��״̬���� (�����)��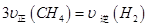
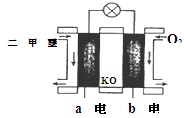
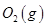 ��KOH(aq)��Ƴ���ͼb��ʾ�ĵ��װ�ã���õ�ظ����ĵ缫��ӦʽΪ ��
��KOH(aq)��Ƴ���ͼb��ʾ�ĵ��װ�ã���õ�ظ����ĵ缫��ӦʽΪ ��
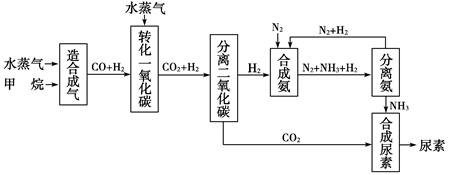
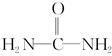 ����д�����ֺ���̼��˫�������ص�ͬ���칹��Ľṹ��ʽ��
����д�����ֺ���̼��˫�������ص�ͬ���칹��Ľṹ��ʽ��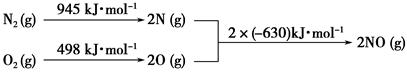

 CH3OCH3��H2O
CH3OCH3��H2O CH3OCH3(g)��CO2(g) ��H����247kJ/mol
CH3OCH3(g)��CO2(g) ��H����247kJ/mol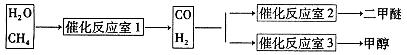

 3AlCl(g)+3CO(g)����H="a" kJ��mol-1
3AlCl(g)+3CO(g)����H="a" kJ��mol-1