��Ŀ����
X��Y��Z��WΪ������ͬ�������ķ��ӻ����ӣ�����ԭ������С��10��Ԫ����ɣ�X��5��ԭ�Ӻˡ�ͨ��״���£�WΪ��ɫҺ�塣
��֪:X+Y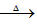 Z+W
Z+W
(1)Y�ĵ���ʽ��_________________________��
(2)Һ̬Z��W�ĵ������ƣ����ɵ������������ͬ���������ӣ�Һ̬Z�ĵ��뷽��ʽ��_________________________________��
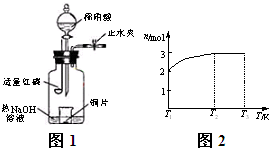
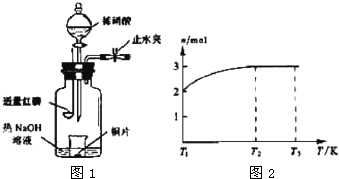
(3)��ͼʾװ���Ʊ�NO����֤�仹ԭ�ԡ���������Ҫ������
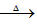 Z+W
Z+W (1)Y�ĵ���ʽ��_________________________��
(2)Һ̬Z��W�ĵ������ƣ����ɵ������������ͬ���������ӣ�Һ̬Z�ĵ��뷽��ʽ��_________________________________��
(3)��ͼʾװ���Ʊ�NO����֤�仹ԭ�ԡ���������Ҫ������

a.����ƿ��ע��������NaOH��Һ����ʢ��ͭƬ��С�ձ�����ƿ�С�
b.�ر�ֹˮ�У���ȼ���ף�����ƿ�У����ý�����
c.�����׳��ȼ�գ�һ��ʱ����Һ©�����������ձ��е�������ϡ���ᡣ
�ٲ���c��ȱ�ٵ�һ����Ҫ������__________________________��
�ں��׳��ȼ�յIJ�����NaOH��Һ��Ӧ�����ӷ���ʽ��__________________��
�۲���c����ϡ������ձ��е�������________________________����Ӧ�����ӷ���ʽ��__________________��
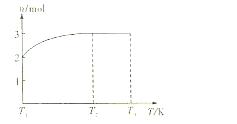
(4)һ���¶��£���1 mol N2O4�����ܱ������У�����ѹǿ���䣬�����¶���T1�Ĺ����У���������ɫ��Ϊ����ɫ���¶���T1�������ߵ�T2�Ĺ����У�������Ϊ��ɫ��������T2������ѹǿ��������Ϊ����ɫ����������ʵ���n���¶�T�仯�Ĺ�ϵ��ͼ��ʾ��
b.�ر�ֹˮ�У���ȼ���ף�����ƿ�У����ý�����
c.�����׳��ȼ�գ�һ��ʱ����Һ©�����������ձ��е�������ϡ���ᡣ
�ٲ���c��ȱ�ٵ�һ����Ҫ������__________________________��
�ں��׳��ȼ�յIJ�����NaOH��Һ��Ӧ�����ӷ���ʽ��__________________��
�۲���c����ϡ������ձ��е�������________________________����Ӧ�����ӷ���ʽ��__________________��
(4)һ���¶��£���1 mol N2O4�����ܱ������У�����ѹǿ���䣬�����¶���T1�Ĺ����У���������ɫ��Ϊ����ɫ���¶���T1�������ߵ�T2�Ĺ����У�������Ϊ��ɫ��������T2������ѹǿ��������Ϊ����ɫ����������ʵ���n���¶�T�仯�Ĺ�ϵ��ͼ��ʾ��
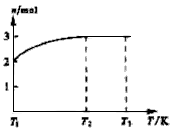
���¶���T1-T2֮�䣬��Ӧ�Ļ�ѧ����ʽ��_________________________��
���¶���T2-T3֮�䣬�����ƽ����Է��������ǣ�����1λС����______________��
���¶���T2-T3֮�䣬�����ƽ����Է��������ǣ�����1λС����______________��
(1)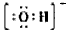
(2)2NH3(l) NH2-+NH4+
NH2-+NH4+
(3) �ٴ�ֹˮ�У�ͨ��������������P2O5+6OH-=2PO43-+3H2O����CuƬ���ܽ⣬����ɫ���ݲ�������Һ����ɫ��Ϊ��ɫ��3Cu+8H++2NO3-=3Cu2++2NO��+4H2O
(4) ��2NO2 N2O4����30.7
N2O4����30.7
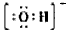
(2)2NH3(l)
 NH2-+NH4+
NH2-+NH4+ (3) �ٴ�ֹˮ�У�ͨ��������������P2O5+6OH-=2PO43-+3H2O����CuƬ���ܽ⣬����ɫ���ݲ�������Һ����ɫ��Ϊ��ɫ��3Cu+8H++2NO3-=3Cu2++2NO��+4H2O
(4) ��2NO2
 N2O4����30.7
N2O4����30.7 
��ϰ��ϵ�д�
 ÿ�α���ϵ�д�
ÿ�α���ϵ�д� ��ѧ����ϵ�д�
��ѧ����ϵ�д�
�����Ŀ
 X��Y��Z��WΪ������ͬ�������ķ��ӻ����ӣ�����ԭ������С��10��Ԫ����ɣ�X��5��ԭ�Ӻˣ�ͨ��״���£�WΪ��ɫҺ�壮��֪��X+Y
X��Y��Z��WΪ������ͬ�������ķ��ӻ����ӣ�����ԭ������С��10��Ԫ����ɣ�X��5��ԭ�Ӻˣ�ͨ��״���£�WΪ��ɫҺ�壮��֪��X+Y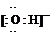
 ��2008?������X��Y��Z��WΪ������ͬ�������ķ��ӻ����ӣ�����ԭ������С��10��Ԫ����ɣ�X��5��ԭ�Ӻˣ�ͨ��״���£�WΪ��ɫҺ�壮
��2008?������X��Y��Z��WΪ������ͬ�������ķ��ӻ����ӣ�����ԭ������С��10��Ԫ����ɣ�X��5��ԭ�Ӻˣ�ͨ��״���£�WΪ��ɫҺ�壮
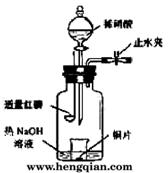 (3)��ͼʾװ���Ʊ�NO����֤�仹ԭ�ԡ���������Ҫ������
(3)��ͼʾװ���Ʊ�NO����֤�仹ԭ�ԡ���������Ҫ������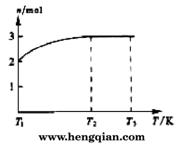 (4)һ���¶��£���1 mol N2O4�����ܱ������У�����ѹǿ���䣬�����¶���T1�Ĺ����У���������ɫ��Ϊ����ɫ���¶���T1�������ߵ�T2�Ĺ����У�������Ϊ��ɫ��������T2,����ѹǿ��������Ϊ����ɫ����������ʵ���n���¶�T�仯�Ĺ�ϵ��ͼ��ʾ��
(4)һ���¶��£���1 mol N2O4�����ܱ������У�����ѹǿ���䣬�����¶���T1�Ĺ����У���������ɫ��Ϊ����ɫ���¶���T1�������ߵ�T2�Ĺ����У�������Ϊ��ɫ��������T2,����ѹǿ��������Ϊ����ɫ����������ʵ���n���¶�T�仯�Ĺ�ϵ��ͼ��ʾ��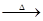 Z+W
Z+W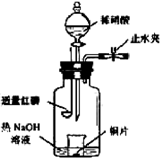
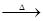 Z+W
Z+W