��Ŀ����
����Ŀ���±���Ԫ�����ڱ���һ���֣���Ա��еĢ١�����Ԫ�أ���д���пհ�(��д��Ų��÷�)
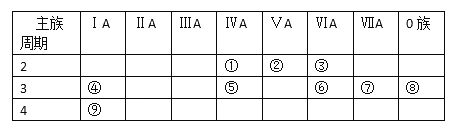
��1��������������ˮ�����У�������ǿ�Ļ�����Ļ�ѧʽ��________��
��2���ȽϢ���ݵ�����������Ӧ��ˮ�������ǿ����________���ѧʽ������ͨ��________��д��Ӧ�Ļ�ѧ����ʽ��˵����
��3��ʵ������ȡ�ڵ��⻯��Ļ�ѧ����ʽ________
��4���ڿ����γɶ������������һ���Ǻ���ɫ���壬���÷���ʽ˵�������岻�˲�����ˮ���ռ���ԭ��________���û�ѧ����ʽ��ʾ��
��5���ȽϢ�����⻯����ȶ�________���ѧʽ��
��6���ۢ���Ԫ����ɵĻ�����A2B2�͵���ʽΪ��________�����ڵĻ�ѧ����________��д��A2B2��ˮ��Ӧ�Ļ�ѧ����ʽ______________��
��7����ĵ�����ˮ��Ӧ�������ӻ������������Ӱ뾶_______S2-���>������)
���𰸡� HClO4 H2CO3 H2CO3+Na2SiO3=Na2CO3+H2SiO3�� Ca(OH)2+2NH4Cl![]() CaCl2+3NH3��+2H2O 2NO2+H2O=2HNO3+NO H2O
CaCl2+3NH3��+2H2O 2NO2+H2O=2HNO3+NO H2O ![]() ���ۼ������Ӽ� 2Na2O2+2H2O=4NaOH+O2�� <
���ۼ������Ӽ� 2Na2O2+2H2O=4NaOH+O2�� <
����������Ԫ�������ڱ��е�λ�ÿ�֪����ΪC����ΪN����ΪO����ΪNa����ΪSi����ΪS����ΪCl����ΪAr����ΪK��
(1)����Ԫ����Cl����ۺ�����--������������ǿ�������ữѧʽΪHClO4���ʴ�Ϊ��HClO4��
(2)ͬ�������϶��£��ǽ����Լ���������������Ӧˮ��������Լ�����������H2CO3��H2SiO3����������ǿ���Ʊ�����ԭ��������֤������ʽΪ��Na2SiO3+CO2+H2O=H2SiO3��+Na2CO3���ʴ�Ϊ��H2CO3��Na2SiO3+CO2+H2O=H2SiO3��+Na2CO3��
(3)ʵ�������Ȼ�����������Ʒ�Ӧ�Ʊ���������Ӧ�����Ȼ��ơ�������ˮ����Ӧ����ʽΪ��2NH4Cl+Ca(OH)2![]() CaCl2+2NH3��+2H2O���ʴ�Ϊ��2NH4Cl+Ca(OH)2
CaCl2+2NH3��+2H2O���ʴ�Ϊ��2NH4Cl+Ca(OH)2![]() CaCl2+2NH3��+2H2O��
CaCl2+2NH3��+2H2O��
(4)�ú���ɫ����Ϊ��������������������ˮ��Ӧ����������NO����Ӧ����ʽΪ��3NO2+H2O=2HNO3+NO���ʶ���������������ˮ���ռ����ʴ�Ϊ��3NO2+H2O=2HNO3+NO��
(5)ͬ�������϶��·ǽ����Լ�������̬�⻯���ȶ��Լ�������H2O���ȶ����ʴ�Ϊ��H2O��
(6)�ۢ���Ԫ����ɵĻ�����Na2O2��������������������ӹ��ɣ�����ʽΪ![]() ���������Ӽ������ۼ�������������ˮ��Ӧ����������������������Ӧ����ʽΪ��2Na2O2+2H2O=4NaOH+O2�����ʴ�Ϊ��
���������Ӽ������ۼ�������������ˮ��Ӧ����������������������Ӧ����ʽΪ��2Na2O2+2H2O=4NaOH+O2�����ʴ�Ϊ��![]() �����Ӽ������ۼ���2Na2O2+2H2O=4NaOH+O2����
�����Ӽ������ۼ���2Na2O2+2H2O=4NaOH+O2����
(7)��ĵ�����ˮ��Ӧ�������ӻ�����ΪKOH�����е�������ΪK+�����Ӳ�ṹ��ͬ�����ӣ��˵����Խ�����Ӱ뾶ԽС�������Ӱ뾶K+��S2-���ʴ�Ϊ������

 ��ͼͼ�麮����ҵ������ҵ���ִ�ѧ������ϵ�д�
��ͼͼ�麮����ҵ������ҵ���ִ�ѧ������ϵ�д�����Ŀ��ʵ�������÷�ͭ�Ͻ�(������������)����ȡ����ͭ����(CuSO4��5H2O)���������£�

�������������������������pH�ɲο��������ݣ�
Fe3�� | Fe2�� | Cu2�� | Al3�� | |
��ʼ����ʱ��pH | 2.2 | 7.5 | 5.2 | 3.7 |
��ȫ����ʱ��pH | 3.2 | 9.0 | 6.7 | 4.7 |
��ش�
��1�����ܽ�Ͻ�ʱ����Ļ�����2 L 3 mol��L��1�����1 L 2 mol��L��1�����϶��ɣ���Ӧ�����ɱ�״���µ�NO��������Ϊ____________L��
��2������H2O2��Ŀ����____________��
��3��Ϊ��֤��Ʒ�Ĵ��ȣ�M�������ѡ��________(����ĸ)������pH�ķ�ΧΪ____________��
a��Cu(OH)2��b��H2SO4��c��NH3��H2O��d��Na2CO3
��4����ҺD�м��������Ŀ��Ϊ_______________________________________��
��5������ҺE�Ƶ�CuSO4��5H2O��������Ҫ�IJ���Ϊ________���ᾧ�����˺��
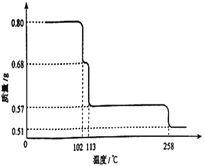
��6��0.80g CuSO4��5H2O��Ʒ������ˮ���̵��������ߣ���Ʒ�������¶ȱ仯�����ߣ���ͼ��ʾ����ȷ��200��ʱ�������ʵĻ�ѧʽ______________��
����Ŀ���������ʵ��׳ơ��ɷּ���;��Ӧ��ȷ���ǣ� ��
A | B | C | D | |
�׳� | Ư�۾� | С�մ� | ���� | ���� |
�ɷ� | NaClO | Na2CO3 | BaCO3 | Fe2O3 |
��; | ������ | ���ͷ� | X��Ƭ | Ϳ�� |
A. A B. B C. C D. D