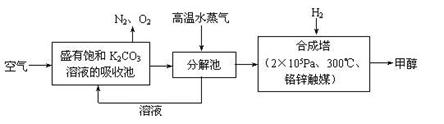
��Ŀ����
(1)�µġ�����������������(GB 3095 2012)����2016��1��1�����ҹ�ȫ��ʵʩ���ݴ�,������������ָ��(AQI)�ձ���ʵʱ���������SO2��NO2��CO��O3��PM10��PM2.5��ָ��,Ϊ�����ṩ����ָ��,�������ؾ���������ų��к����
2012)����2016��1��1�����ҹ�ȫ��ʵʩ���ݴ�,������������ָ��(AQI)�ձ���ʵʱ���������SO2��NO2��CO��O3��PM10��PM2.5��ָ��,Ϊ�����ṩ����ָ��,�������ؾ���������ų��к����
�������ų���β���к���CO��NO������,�û�ѧ����ʽ���Ͳ���NO��ԭ�� ��
�������������ڰ�װ�Ĵ�ת����,��ʹ����β���е���Ҫ��Ⱦ��ת��Ϊ���Ĵ���ѭ�����ʡ���֪:
N2(g)+O2(g)="2NO(g)" ��H="+180.5" kJ/mol
2C(s)+O2(g)="2CO(g)" ��H="-221.0" kJ/mol
C(s)+O2(g)=CO2(g) ��H="-393.5" kJ/mol
��Ӧ2NO(g)+2CO(g)=N2(g)+2CO2(g)�Ħ�H= kJ/mol��
(2)ֱ���ŷŵ���������γ����ꡢ����,����ԭ�����������շ��dz��õĴ�������������NH3��CH4�������ȥ�����еĵ��������֪:CH4(g)+2O2(g)=CO2(g)+2H2O(l) ��H1="a" kJ/mol;�����㷴ӦCH4(g)+4NO(g)=CO2(g)+2H2O(l)+2N2(g)���ʱ䦤H2����Ҫ��ѯij��Ӧ���ʱ䦤H3,����Ӧ�и����ʵĻ�ѧ������֮��Ϊ���������ʱ,��H3="b" kJ/mol,�÷�Ӧ���Ȼ�ѧ����ʽ�� ,�ݴ˼������H2= kJ/mol(�ú�a��b��ʽ�ӱ�ʾ)��
(3)�±��г��˹�ҵ������SO2�����ַ�����
��������Ҫ���������з�Ӧ:
2CO(g)+SO2(g)=S(g)+2CO2(g) ��H="+8.0" kJ/mol
2H2(g)+SO2(g)=S(g)+2H2O(g)��H="+90.4" kJ/mol
2CO(g)+O2(g)=2CO2(g) ��H="-566.0" kJ/mol
2H2(g)+O2(g)=2H2O(g) ��H="-483.6" kJ/mol
��S(g)��O2(g)��Ӧ����SO2(g)���Ȼ�ѧ����ʽ�ɱ�ʾΪ ��
(4)�ϳɰ��õ��������Լ���Ϊԭ���Ƶá��йػ�ѧ��Ӧ�������仯��ͼ��ʾ,��CH4(g)��H2O(g)��Ӧ����CO(g)��H2(g)���Ȼ�ѧ����ʽΪ ��



 2012)����2016��1��1�����ҹ�ȫ��ʵʩ���ݴ�,������������ָ��(AQI)�ձ���ʵʱ���������SO2��NO2��CO��O3��PM10��PM2.5��ָ��,Ϊ�����ṩ����ָ��,�������ؾ���������ų��к����
2012)����2016��1��1�����ҹ�ȫ��ʵʩ���ݴ�,������������ָ��(AQI)�ձ���ʵʱ���������SO2��NO2��CO��O3��PM10��PM2.5��ָ��,Ϊ�����ṩ����ָ��,�������ؾ���������ų��к�����������ų���β���к���CO��NO������,�û�ѧ����ʽ���Ͳ���NO��ԭ�� ��
�������������ڰ�װ�Ĵ�ת����,��ʹ����β���е���Ҫ��Ⱦ��ת��Ϊ���Ĵ���ѭ�����ʡ���֪:
N2(g)+O2(g)="2NO(g)" ��H="+180.5" kJ/mol
2C(s)+O2(g)="2CO(g)" ��H="-221.0" kJ/mol
C(s)+O2(g)=CO2(g) ��H="-393.5" kJ/mol
��Ӧ2NO(g)+2CO(g)=N2(g)+2CO2(g)�Ħ�H= kJ/mol��
(2)ֱ���ŷŵ���������γ����ꡢ����,����ԭ�����������շ��dz��õĴ�������������NH3��CH4�������ȥ�����еĵ��������֪:CH4(g)+2O2(g)=CO2(g)+2H2O(l) ��H1="a" kJ/mol;�����㷴ӦCH4(g)+4NO(g)=CO2(g)+2H2O(l)+2N2(g)���ʱ䦤H2����Ҫ��ѯij��Ӧ���ʱ䦤H3,����Ӧ�и����ʵĻ�ѧ������֮��Ϊ���������ʱ,��H3="b" kJ/mol,�÷�Ӧ���Ȼ�ѧ����ʽ�� ,�ݴ˼������H2= kJ/mol(�ú�a��b��ʽ�ӱ�ʾ)��
(3)�±��г��˹�ҵ������SO2�����ַ�����
| ������ | �ð�ˮ��SO2ת��(NH4)2SO3,��������(NH4)2SO4 |
| ������ | ���������Ƚ���(��Ҫ�ɷ�CO��CH4��H2)��SO2�ڸ����»�ԭ�ɵ����� |
| ������ | ��Na2SO3��Һ����SO2,�پ����ת��ΪH2SO4 |
2CO(g)+SO2(g)=S(g)+2CO2(g) ��H="+8.0" kJ/mol
2H2(g)+SO2(g)=S(g)+2H2O(g)��H="+90.4" kJ/mol
2CO(g)+O2(g)=2CO2(g) ��H="-566.0" kJ/mol
2H2(g)+O2(g)=2H2O(g) ��H="-483.6" kJ/mol
��S(g)��O2(g)��Ӧ����SO2(g)���Ȼ�ѧ����ʽ�ɱ�ʾΪ ��
(4)�ϳɰ��õ��������Լ���Ϊԭ���Ƶá��йػ�ѧ��Ӧ�������仯��ͼ��ʾ,��CH4(g)��H2O(g)��Ӧ����CO(g)��H2(g)���Ȼ�ѧ����ʽΪ ��



(1)��N2+O2 2NO ��-746.5
2NO ��-746.5
(2)N2(g)+O2(g)="2NO(g)" ��H3="b" kJ/mol a-2b
(3)S(g)+O2(g)=SO2(g) ��H="-574.0" kJ/mol
(4)CH4(g)+H2O(g)=CO(g)+3H2(g)��H="+161.1" kJ/mol
 2NO ��-746.5
2NO ��-746.5(2)N2(g)+O2(g)="2NO(g)" ��H3="b" kJ/mol a-2b
(3)S(g)+O2(g)=SO2(g) ��H="-574.0" kJ/mol
(4)CH4(g)+H2O(g)=CO(g)+3H2(g)��H="+161.1" kJ/mol
(1)�ڰ����˳���3���Ȼ�ѧ����ʽ���,�ɸ�˹���ɢۡ�2-��-�ڵ�2NO(g)+2CO(g) N2(g)+2CO2(g)�Ħ�H="[-393.5��2-180.5-(-221.0)]kJ/mol=-746.5" kJ/mol��
N2(g)+2CO2(g)�Ħ�H="[-393.5��2-180.5-(-221.0)]kJ/mol=-746.5" kJ/mol��
(2)���ݸ�˹���ɽ���֪�������Ȼ�ѧ����ʽ����ɵ��Ȼ�ѧ����ʽΪ2N2(g)+2O2(g)="4NO(g)" ��H="a" kJ/mol-��H2="2b" kJ/mol,��H2=(a-2b)kJ/mol,ͬʱ������ķ���ʽ����H������2�ɵ��Ȼ�ѧ����ʽ��
(3)���������������ʽ��Ӽ�ȥǰ��������ʽ�ٳ���2�ɵ���Ӧ���Ȼ�ѧ����ʽ:S(g)+O2(g)=SO2(g)��H="-574.0" kJ/mol��
(4)�����˳���ͼ���е�3���Ȼ�ѧ����ʽ���,���ݸ�˹����,��-�ڡ�3-�ٵ�CH4(g)+H2O(g)=CO(g)+3H2(g)��H="[-846.3-(-241.8)��3-(-282.0)]kJ/mol=+161.1" kJ/mol��
 N2(g)+2CO2(g)�Ħ�H="[-393.5��2-180.5-(-221.0)]kJ/mol=-746.5" kJ/mol��
N2(g)+2CO2(g)�Ħ�H="[-393.5��2-180.5-(-221.0)]kJ/mol=-746.5" kJ/mol��(2)���ݸ�˹���ɽ���֪�������Ȼ�ѧ����ʽ����ɵ��Ȼ�ѧ����ʽΪ2N2(g)+2O2(g)="4NO(g)" ��H="a" kJ/mol-��H2="2b" kJ/mol,��H2=(a-2b)kJ/mol,ͬʱ������ķ���ʽ����H������2�ɵ��Ȼ�ѧ����ʽ��
(3)���������������ʽ��Ӽ�ȥǰ��������ʽ�ٳ���2�ɵ���Ӧ���Ȼ�ѧ����ʽ:S(g)+O2(g)=SO2(g)��H="-574.0" kJ/mol��
(4)�����˳���ͼ���е�3���Ȼ�ѧ����ʽ���,���ݸ�˹����,��-�ڡ�3-�ٵ�CH4(g)+H2O(g)=CO(g)+3H2(g)��H="[-846.3-(-241.8)��3-(-282.0)]kJ/mol=+161.1" kJ/mol��

��ϰ��ϵ�д�
�����Ŀ
 2NH3����Ӧ��1mol N2�ų�������Ϊ92.4kJ����Ͽ�1mol N��N�������յ������ǣ� ��
2NH3����Ӧ��1mol N2�ų�������Ϊ92.4kJ����Ͽ�1mol N��N�������յ������ǣ� �� 2CO(g)����H="-221.0" kJ��mol-1
2CO(g)����H="-221.0" kJ��mol-1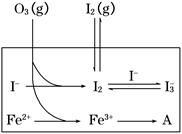
 HOI(aq)����H2
HOI(aq)����H2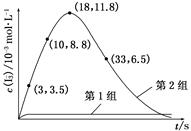
 2SO3(g)��H="-197" kJ��mol-1��ʵ����4 mol SO2�μ�������Ӧ�ų�354 kJ��������SO2��ת������ӽ���( )
2SO3(g)��H="-197" kJ��mol-1��ʵ����4 mol SO2�μ�������Ӧ�ų�354 kJ��������SO2��ת������ӽ���( ) O2(g)
O2(g) H2O(l)��H="-285.8" kJ/mol
H2O(l)��H="-285.8" kJ/mol