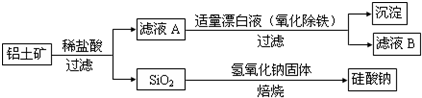
��Ŀ����
11�����������˵��2HI��g��?H2��g��+I2��g���Ѵ�ƽ��״̬���Ǣڢޢᣮ�ٵ�λʱ��������nmolH2��ͬʱ������nmolHI��
��1��H-H�����ѵ�ͬʱ��2��H-I�����ѣ�
�ۻ�����аٷ����ΪHI%=I2%��
�ܷ�Ӧ����v��H2��=v��I2��=$\frac{1}{2}$ v��HI��ʱ��
�ݻ��������c��HI����c��H2����c��I2��=2��1��1ʱ��
���¶Ⱥ����һ��ʱ��ijһ������Ũ�Ȳ��ٱ仯��
���¶Ⱥ����һ��ʱ��������ѹǿ���ٱ仯��
������һ������������ƽ�������������ٱ仯��
���¶Ⱥ����һ��ʱ������������ɫ���ٱ仯��
���¶Ⱥ�ѹǿһ��ʱ�����������ܶȲ��ٱ仯��
�����ޡ����˵������˵��2NO2?N2O4�ﵽƽ��״̬���Ǣޢߢ��⣮
���� ���ݻ�ѧƽ��״̬��������𣬵���Ӧ�ﵽƽ��״̬ʱ�����淴Ӧ������ȣ������ʵ�Ũ�ȡ��ٷֺ������䣬�Լ��ɴ�������һЩ��Ҳ�������仯������ʱҪע�⣬ѡ���жϵ������������ŷ�Ӧ�Ľ��з����仯�������������ɱ仯����ֵʱ��˵�����淴Ӧ����ƽ��״̬�����������
��� �⣺�ٵ�λʱ��������nmolH2��ͬʱ������nmolHI���仯��֮�Ȳ����ڼ�����֮�ȣ��ʴ���
��1��H-H�����ѣ���Ч��2��H-I���γ�ͬʱ��2��H-I�����ѣ����淴Ӧ������ȣ���ƽ��״̬������ȷ��
�۰ٷֺ������䣬��ƽ��ʱ������аٷ���ɿ���ΪHI%=I2%��������ʵij�ʼŨ�ȼ�ת�����йأ��ʴ���
��ֻҪ��Ӧ�������з�Ӧ����v��H2��=v��I2��=$\frac{1}{2}$ v��HI�����ʴ���
�ݵ���ϵ��ƽ��״̬ʱ�����������c��HI����c��H2����c��I2��������2��1��1��Ҳ���ܲ��ǣ�������ʵij�ʼŨ�ȼ�ת�����йأ��ʴ���
���¶Ⱥ����һ��ʱ��ijһ������Ũ�Ȳ��ٱ仯������������ȴ�ƽ��״̬������ȷ��
���¶Ⱥ����һ��ʱ������������ȣ�������ѹǿʼ�ղ��ٱ仯���ʴ���
������һ��������������ȣ���������ƽ����������ʼ�ղ��ٱ仯���ʴ���
���¶Ⱥ����һ��ʱ������������ɫ���ٱ仯��˵���������������䣬��Ӧ��ƽ��״̬������ȷ��
���¶Ⱥ�ѹǿһ��ʱ����������ļ�������ȣ������һ����ֵ�����������ܶ�ʼ�ղ��ٱ仯���ʴ���ѡ�ڢޢᣮ
�����ޡ����˵������˵��2NO2?N2O4�ﵽƽ��״̬���ǣ�
���¶Ⱥ����һ��ʱ��ijһ������Ũ�Ȳ��ٱ仯������������ȴ�ƽ��״̬������ȷ��
���¶Ⱥ����һ��ʱ��������������ȣ�������ѹǿ���ٱ仯��˵����Ӧ��ƽ��״̬������ȷ��
������һ������������ƽ�������������ٱ仯��˵�����ʵ������䷴Ӧ��ƽ��״̬������ȷ��
���¶Ⱥ����һ��ʱ������������ɫ���ٱ仯��˵�����������������䣬��Ӧ��ƽ��״̬������ȷ��
���¶Ⱥ�ѹǿһ��ʱ�����������ܶȲ��ٱ仯��˵����������ʵ������䣬��Ӧ��ƽ��״̬������ȷ��
��ѡ���ޢߢ��⣮
���� ���⿼���˻�ѧƽ��״̬���жϣ��ѶȲ���ע���Ӧ�ﵽƽ��״̬ʱ�����淴Ӧ������ȣ�����Ϊ0��

 ������ϵ�д�
������ϵ�д�| A�� | ��״���£�18 g H2O�к��е�������Ϊ10 NA | |
| B�� | ��״���£�2.24 L C12ͨ��ˮ�У���Ӧת�Ƶĵ�����Ϊ0.1 NA | |
| C�� | ���³�ѹ�£�53.5 g NH4Cl�����к��й��ۼ�����ĿΪ4 NA | |
| D�� | ���³�ѹ�£�2 L 0.5 mol•L-1�������Һ��������������������3 NA |
| A�� | FeCl3 | B�� | NaHCO3 | C�� | MgSO4 | D�� | Na2SO3 |
| A�� | ��״���£�22.4 L���Ȼ�̼���еķ�����ΪNA | |
| B�� | ���³�ѹ�£���CO2ͨ������Na2O2����������������28g���μӷ�Ӧ��CO2������ΪNA | |
| C�� | 0.5 mol•L-1 NaOH��Һ��Na+��Ϊ0.5NA | |
| D�� | ���³�ѹ�£�16 g��������������ΪNA |
| A�� | ����CO2���ŷţ����Լ�������IJ��� | |
| B�� | Ϊ����SO2���ŷţ�����ȼú�м���ʯ��ʯ���������ԴӸ������������� | |
| C�� | ���칬һ�š�ʹ�õ�̼��ά����һ�������л��߷��Ӳ��� | |
| D�� | ��ʳƷ���з���ʢ�й轺�����۵���С�����ɷ�ֹʳ���ܳ����������� |
| ��ѧʽ | CH3COOH | HClO | H2CO3 | H2C2O4 |
Ka | Ka=1.8��10-5 | Ka=3.0��10-8 | Ka1=4.1��10-7 Ka2=5.6��10-11 | Ka1=5.9��10-2 Ka2=6.4��10-5 |
��2��pH ��ͬ��NaClO��CH3COOK��Һ������Һ�����ʵ���Ũ�ȵĴ�С��ϵ�ǣ�CH3COONa��NaClO������Һ�У�[c��Na+��-c��ClO-��]=[c��K+��-c��CH3COO-��]���������������=������
��3���� 0.1mol/LCH3COOH��Һ�еμ�NaOH��Һ��c��CH3COOH����c��CH3COO-��=5��9����ʱ��Һ pH=5��
��4��̼������Һ�еμ�������ˮ�����ӷ���ʽΪCl2+2CO32-+H2O�T2HCO3-+Cl-+ClO-��