��Ŀ����
���������������ش����⡣��Դ�ɻ���Ϊһ����Դ�Ͷ�����Դ����Ȼ�������ֳ���ʽ�ṩ����Դ��Ϊһ����Դ��������������Դ�����������ȡ����Դ��Ϊ������Դ��������һ�ָ�Ч��û����Ⱦ�Ķ�����Դ������������Ȼ���д������ڵ�ˮ����ȡ��
2H2O��l��=2H2��g��+O2��g������H=
+517.6kJ?mol-1
��1������������ȷ���ǣ� ����
A.�����Ƕ�����Դ B.ˮ���Ƕ�����Դ
C.��Ȼ����һ����Դ D.��¯����һ����Դ
��2����֪��CH4��g��+2O2��g����2H2O��l��+CO2��g������H=
-890.3kJ?mol-1��1gH2��1gCH4�ֱ�ȼ�պų�������֮��Լ�ǣ� ����
A.1��3.4 B.1��1.7 C.2.3��1 D.4.6��1
��3��������ˮ��ȡ������ԴH2�������о�������ȷ���ǣ� ����
A.����ˮ����������ǿ���ȼ�յ����ʣ���ˣ����о���ˮ���ֽ������£�ʹ���Ϊ������Դ
B.�跨��̫����۽����������£�ʹˮ�ֽ��������
C.Ѱ�Ҹ�Ч������ʹˮ�ֽ����������ͬʱ�ͷ�����
D.Ѱ�����⻯ѧ���ʣ����ڿ���������Դ���Էֽ�ˮ��ȡ����
��1��AC
��2��C
��3��BD
�����������

��ϰ��ϵ�д�
 ״Ԫ��ȫ��ͻ�Ƶ�����ϵ�д�
״Ԫ��ȫ��ͻ�Ƶ�����ϵ�д�
�����Ŀ
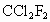 )���ڹ�������·ֽ⣬����Clԭ�ӣ�Clԭ�ӻ�Գ�����������õ��ƻ�����(�����ķ���ʽΪ
)���ڹ�������·ֽ⣬����Clԭ�ӣ�Clԭ�ӻ�Գ�����������õ��ƻ�����(�����ķ���ʽΪ )��
)��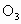
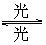

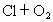
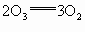
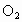 ��
��