��Ŀ����
��������������ȡȾ�Ϻ�һЩҩ�����Ҫ�м��壬���ɱ����ǻ��õ���
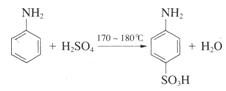
ʵ���ҿ�������ͼʵ��װ�úϳɶ��������ᡣʵ�鲽�����£�
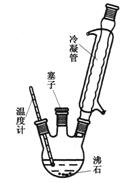
�� ��һ��250 mL������ƿ�м���10 mL�� ����������ʯ��
����������ʯ��
��������ƿ������ˮ����ȴ��С�ĵؼ���18 mLŨ���ᡣ
�� ��������ƿ������ԡ�л���������170��180�棬ά�ִ��¶�2��2.5 h��
�� ����ӦҺ��ȴ��Լ50�����ʢ��100 mL��ˮ���ձ��У��ò��������Ͻ��裬��ʹ�������������ˣ���������ˮϴ�ӣ��õ��ľ����Ƕ���������ֲ�Ʒ��
�� ���ֲ�Ʒ�÷�ˮ�ܽ⣬��ȴ�ᾧ������Һ��ɫ������û���̿��ɫ�������ˣ��ռ���Ʒ�����ɡ���˵����100 mLˮ��20��ʱ���ܽ����������1.08 g����100��ʱ���ܽ�6.67 g����
�Իش���ա�
��1��װ���������ܵ������� �� ��
��2��������в�����ԡ���ȣ�����˵����ȷ���� �� ������ţ���
A������ԡ���ȵĺô��Ƿ�Ӧ�����Ⱦ��ȣ����ڿ����¶�
B���˴�Ҳ���Ը���ˮԡ����
C��ʵ��װ���е��¶ȼƿ��Ըı�λ�ã�Ҳ��ʹ��ˮ�������������
��3���������������ˮϴ�Ӿ���ĺô��� �� ��
��4������ۺܾ͢����г��˲������ڳ������ֹͣ����ʱ��Ӧע���� �� ��
Ȼ�� �� ���Է�������
��5�����������ʱ��Ҫ�����ֲ�Ʒ�÷�ˮ�ܽ⣬��ȴ�ᾧ�����ˡ��IJ������ж�Σ���Ŀ���� ��ÿ�γ��˺��Ӧ��ĸҺ�ռ������������ʵ���������Ŀ���� �� ��

ʵ���ҿ�������ͼʵ��װ�úϳɶ��������ᡣʵ�鲽�����£�

�� ��һ��250 mL������ƿ�м���10 mL��
 ����������ʯ��
����������ʯ����������ƿ������ˮ����ȴ��С�ĵؼ���18 mLŨ���ᡣ
�� ��������ƿ������ԡ�л���������170��180�棬ά�ִ��¶�2��2.5 h��
�� ����ӦҺ��ȴ��Լ50�����ʢ��100 mL��ˮ���ձ��У��ò��������Ͻ��裬��ʹ�������������ˣ���������ˮϴ�ӣ��õ��ľ����Ƕ���������ֲ�Ʒ��
�� ���ֲ�Ʒ�÷�ˮ�ܽ⣬��ȴ�ᾧ������Һ��ɫ������û���̿��ɫ�������ˣ��ռ���Ʒ�����ɡ���˵����100 mLˮ��20��ʱ���ܽ����������1.08 g����100��ʱ���ܽ�6.67 g����
�Իش���ա�
��1��װ���������ܵ������� �� ��
��2��������в�����ԡ���ȣ�����˵����ȷ���� �� ������ţ���
A������ԡ���ȵĺô��Ƿ�Ӧ�����Ⱦ��ȣ����ڿ����¶�
B���˴�Ҳ���Ը���ˮԡ����
C��ʵ��װ���е��¶ȼƿ��Ըı�λ�ã�Ҳ��ʹ��ˮ�������������
��3���������������ˮϴ�Ӿ���ĺô��� �� ��
��4������ۺܾ͢����г��˲������ڳ������ֹͣ����ʱ��Ӧע���� �� ��
Ȼ�� �� ���Է�������
��5�����������ʱ��Ҫ�����ֲ�Ʒ�÷�ˮ�ܽ⣬��ȴ�ᾧ�����ˡ��IJ������ж�Σ���Ŀ���� ��ÿ�γ��˺��Ӧ��ĸҺ�ռ������������ʵ���������Ŀ���� �� ��
����12�֣���1������������1�֣�
��2��AC��2�֣��д���0�֣�©ѡ��1�֣�
��3�����ٰ������������ʧ��1�֣�
��4���������ӱú�����ƿ����Ƥ�ܣ�2�֣��ر�ˮ��ͷ��2�֣�
��5����߶���������Ĵ��ȣ�2�֣� ��߰���������IJ��ʣ�2�֣�
��2��AC��2�֣��д���0�֣�©ѡ��1�֣�
��3�����ٰ������������ʧ��1�֣�
��4���������ӱú�����ƿ����Ƥ�ܣ�2�֣��ر�ˮ��ͷ��2�֣�
��5����߶���������Ĵ��ȣ�2�֣� ��߰���������IJ��ʣ�2�֣�
��

��ϰ��ϵ�д�
 �Ͻ�ƽСѧ��������ϵ�д�
�Ͻ�ƽСѧ��������ϵ�д� �Ƹ������������ϵ�д�
�Ƹ������������ϵ�д�
�����Ŀ

 ɫ��___________________________________________��
ɫ��___________________________________________��